Question: Please use the values in the resources listed below instead of the textbook values. Perform the following calculations. (a) Calculate [Cu+] (in M) in
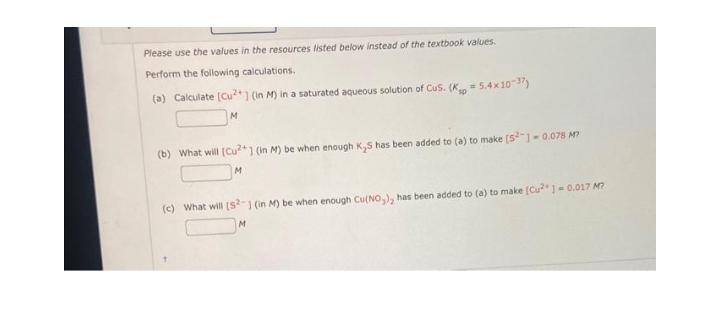
Please use the values in the resources listed below instead of the textbook values. Perform the following calculations. (a) Calculate [Cu+] (in M) in a saturated aqueous solution of CuS. (K 5.4x10-37) M (b) What will [Cu2+] (in M) be when enough K,S has been added to (a) to make [52] -0.078 M7 M (c) What will [52-1 (in M) be when enough Cu(NO), has been added to (a) to make [Cu]=0.017 M? M
Step by Step Solution
3.49 Rating (146 Votes )
There are 3 Steps involved in it
The detailed ... View full answer

Get step-by-step solutions from verified subject matter experts


