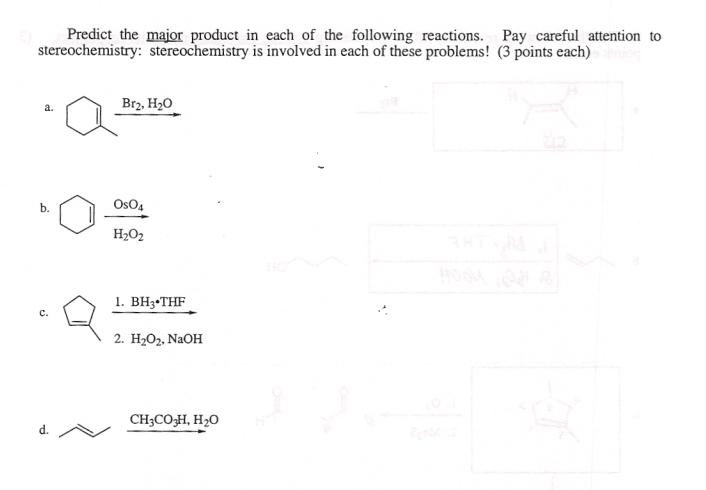
Predict the major product in each of the following reactions. stereochemistry: stereochemistry is involved in each...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Predict the major product in each of the following reactions. stereochemistry: stereochemistry is involved in each of these problems! a. b. d. Br2, H₂O Os04 H₂O₂ 1. BH3-THF 2. H₂O₂, NaOH CH3CO3H, H₂O Pay careful attention to (3 points each) Predict the major product in each of the following reactions. stereochemistry: stereochemistry is involved in each of these problems! a. b. d. Br2, H₂O Os04 H₂O₂ 1. BH3-THF 2. H₂O₂, NaOH CH3CO3H, H₂O Pay careful attention to (3 points each)
Expert Answer:
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Predict the major product in each of the following reactions: CH (a) H20 CHCH-CH3H2CH H2SO4 (Addition of H20 occurs.) (b) CH-CH CH (c) HBr (d) 2 HCI %3 CHCH2CH2CH2CH%3DCH2
-
Predict the predominant product in each of the following reactions. Explain your answer. 3,4-dibromopyridine + NH3, heat (C5H5BrN2)
-
Predict the major organic product in each of the following reactions:
-
North Folk Dairy has an unused bank revolving credit of $50 million. North Folk Dairys vendor provided an invoice in the amount of $100,000 with payment terms of 2/10, net 45. Compute the nominal...
-
Various Reporting formats The following information was taken from the records of Gibson Inc. for the year 2010 Income tax applicable to income from continuing operations $119,000; income tax...
-
For the ideal power cycle shown in Fig. 8.37, assume the steam leaving the boiler at state 5 is at 1200 psia and 1000°F with a mass flux of 50 lbm/s. Steam is removed at state 6 at 400 psia and...
-
On January 1 of each of the first four years of its existence, Allway Company purchases a new unit of equipment. Each unit has a four-year life and zero salvage value, costs \(\$ 100,000\), and is...
-
On December 31, 2008, Ed Abbey Co. performed environmental consulting services for Hayduke Co. Hayduke was short of cash, and Abbey Co. agreed to accept a $200,000 zero-interest-bearing note due...
-
Please make sure the answer isdefinitely correct, and explain how you got it. Thanks!!! Ying Import has several bond issues outstanding. each making semiannual interest payments. The bonds are listed...
-
The isotope technetium-99m is used by doctors to image the skeleton and heart muscles. Fol- lowing injection, the concentration of this isotope decays exponentially in the body. a. Let N(t) represent...
-
In this mini-case you will complete the questionnaire for understanding EarthWear Clothiers, Inc. and the environment in which it operates. INSTRUCTIONS: The worksheet titled "Questionnaire" contains...
-
Globe Travel Agency pays an employee \(\$ 600\) in wages each Friday for a five-day work week ending on that day. The last Friday of January falls on January 27. What adjusting entry should be made...
-
The publisher of International View, a monthly magazine, received two-year subscriptions totaling \(\$ 12,240\) on January 1. (a) What entry should be made to record the receipt of the \(\$ 12,240\)...
-
On January 1, Prepaid Insurance was debited with the cost of a two-year premium in the amount of \(\$ 3,744\). What adjusting entry should be made on January 31 before the January financial...
-
Rally Corps Eastern Division has sales of \($190,000\), cost of goods sold of \($110,000\), operating expenses of \($35,000\), average invested assets of \($900,000\), and a hurdle rate of 12...
-
The table below contains information about five jobs waiting to be processed at stamping machine 307 in the MetalBright Job Shop. a. Which of these three rules provides the best sequence in terms of...
-
1.consider the following table that contains selected data for coca cola Amatill (CCL) for financial year 2018. NET SALES COAST OF SALES Account recivable invent accounts payble current assest...
-
Find the market equilibrium point for the following demand and supply functions. Demand: 2p = - q + 56 Supply: 3p - q = 34
-
The reaction of an aldehyde or ketone with a Grignard reagent is a nucleophilic addition to the carbon-oxygen double bond. (a) What is the nucleophile? (b) The magnesium portion of the Grignard...
-
Give the amino acid sequence of the following polypeptides using only the data given by partial acidic hydrolysis: (a) (b) S, O, P, T H2o STTO PS A, R, C, V, LAC + CR+RV +LA H20
-
Show how you might prepare 2-bromobutane from (a) 2-Butanol (b) 1-Butanol (c) 1-Butene (d) 1-Butyne
-
Prepare a consolidated balance sheet from the following details as at 31 March 2006. During the year, Pa sold goods which had cost 1,900 to Daughter for 2,800. None of these goods had been sold by...
-
The following information relates to Heather Limited and its subsidiary, Thistle*Limited. 1. Heather Limited Retained profits as at 31 March 2008 700,000. 80,000 ordinary shares were purchased in...
-
Draw up a consolidated balance sheet as at 31 December 2007 from the following: At the balance sheet date, Child owes Parent 3,200. During the year Parent sold goods which had cost 6,000 to Child for...

Study smarter with the SolutionInn App



