A copper calorimeter can with mass 0.445 kg contains 0.0900 kg of ice. The system is...
Fantastic news! We've Found the answer you've been seeking!
Question:
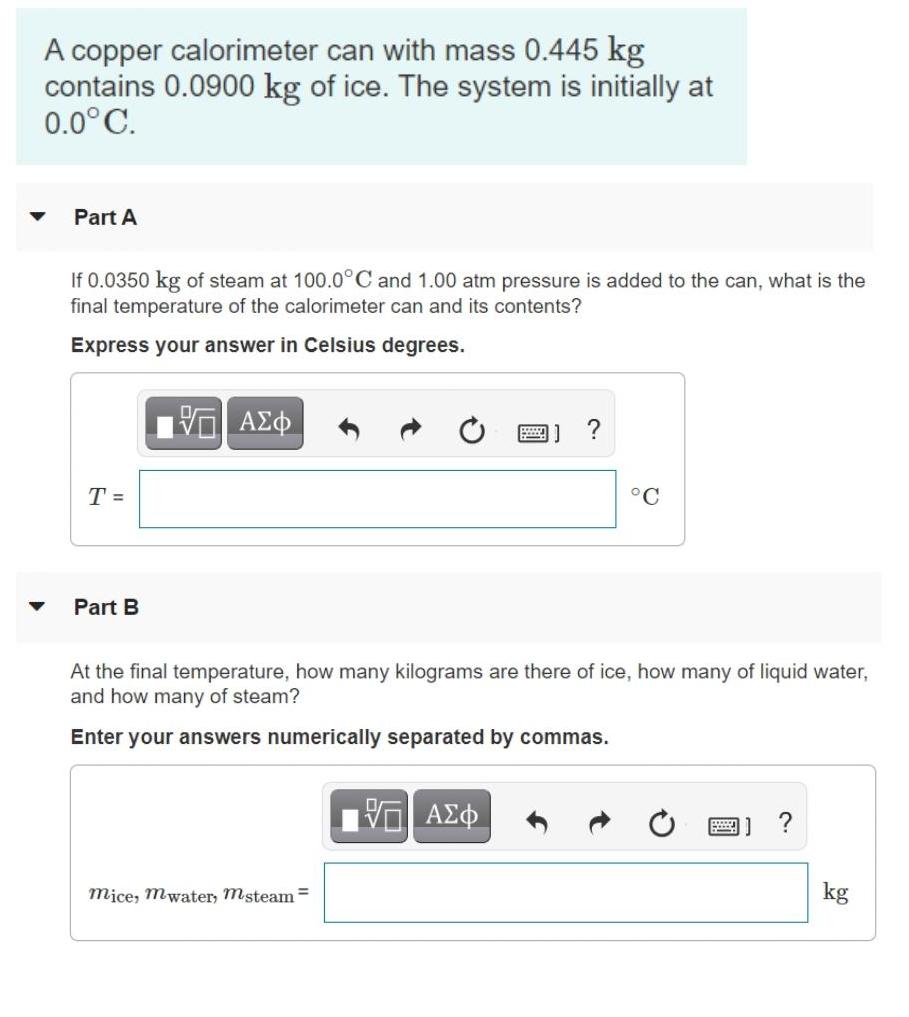
Transcribed Image Text:
A copper calorimeter can with mass 0.445 kg contains 0.0900 kg of ice. The system is initially at 0.0°C. Part A If 0.0350 kg of steam at 100.0°C and 1.00 atm pressure is added to the can, what is the final temperature of the calorimeter can and its contents? Express your answer in Celsius degrees. Π ΑΣΦ ? T = °C Part B At the final temperature, how many kilograms are there of ice, how many of liquid water, and how many of steam? Enter your answers numerically separated by commas. mice, mwater, msteam= kg A copper calorimeter can with mass 0.445 kg contains 0.0900 kg of ice. The system is initially at 0.0°C. Part A If 0.0350 kg of steam at 100.0°C and 1.00 atm pressure is added to the can, what is the final temperature of the calorimeter can and its contents? Express your answer in Celsius degrees. Π ΑΣΦ ? T = °C Part B At the final temperature, how many kilograms are there of ice, how many of liquid water, and how many of steam? Enter your answers numerically separated by commas. mice, mwater, msteam= kg
Expert Answer:

Related Book For 

Engineering Mechanics Dynamics
ISBN: 9781118885840
8th Edition
Authors: James L. Meriam, L. G. Kraige, J. N. Bolton
Posted Date:
Students also viewed these physics questions
-
A copper calorimeter can with mass 0.446 kg contains 0.0950 kg of ice. The system is initially at 0.0oC. (a) If 0.0350 kg of steam at l00.0oC and 1.00 atm pressure is added to the can, what is the...
-
A copper calorimeter can with mass 0.100 kg contains 0.160 kg of water and 0.0180 kg of ice in thermal equilibrium at atmospheric pressure. H 0.750 kg of lead at a temperature of 255C is dropped into...
-
An isolated system is initially at rest. Is it possible for parts of the system to be in motion at some later time? If so, explain how this might occur.
-
Why will a reduction in the real interest rate increase investment spending, other things equal?
-
The null and alternative hypotheses for a one-way ANOVA test are, respectively, H0: 1 = 2 = = k Ha: Not all means are equal. Suppose that, in reality, the null hypothesis is false. Does that mean...
-
Calculating Net Capital Spending Kahne Driving Schools 2006 balance sheet showed net fixed assets of $4.6 million, and the 2007 balance sheet showed net fixed assets of $5.2 million. The companys...
-
The 2005 comparative balance sheet and income statement of Get Wired, Inc., follow on the next page. Get Wired, Inc., had no noncash investing and financing transactions during 2005. During the year,...
-
For the following project, 3 sections are expected every week. Each week is 5 working days with 8 hours a day. Find the value of X1? Find the value of X2? Find the value of X3? Find the value of X4?...
-
5. (20 points) Suppose you deposit $1,000 dollar on Bank of America(BOA) and following T- account reflects the change of balance sheet for this activity. Assets Liabilities Reserves +$1,000 Deposits...
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
What role do neuroplasticity and epigenetic modifications play in shaping an individual's susceptibility to stress-related disorders, and how might targeted interventions capitalize on these...
-
Sally Omar is the manager of the office products division of Pembina Enterprises. In this position, her annual bonus is based on an appraisal of return on investment (ROI) measured as Division income...
-
Jack'sScent Shoppe is the name of a sole proprietorship that carries on a retail business. The business has a December 31 fiscal period and began in2023. In its first fiscal period?business, sales...
-
A woman sings at a fifth harmonic frequency of 3036 Hz. Treat her vocal tract as though it were a closed-open end air column and that the speed of sound is 340 m/s. 1) What would be the frequency of...
-
Factor the expression. 6x3/218x1/2 + 12x-1/2
-
Cohen Company's taxable income this year was $47,000. During the year, Cohen received interest on a municipal bond of $4,000. The company had federal income tax of $6,000 and incurred penalties of...
-
Recall that the set S={1,2,2) is a basis for P2. For p and q polynomials in P define their inner product to be (p. 9) = p()q() at Give an orthogonal basis for P2 with respect to this inner product.
-
Fill in each blank so that the resulting statement is true. A solution to a system of linear equations in two variables is an ordered pair that__________ .
-
If a torque M = Mk is applied to the shaft in Prob. 7/75, determine the x- and y-components of the force supported by the bearing B as the bar and shaft start from rest in the position shown. Neglect...
-
The car C increases its speed at the constant rate of 1.5 m/s 2 as it rounds the curve shown. If the magnitude of the total acceleration of the car is 2.5 m/s 2 at point A where the radius of...
-
Determine I xx for the cylinder with a centered circular hole. The mass of the body is m. r2
-
Consider a bivariate time series \(\boldsymbol{z}_{t}\), where \(z_{1 t}\) is the change in monthly U.S. treasury bills with maturity 3 months and \(z_{2 t}\) is the inflation rate, in percentage, of...
-
Consider the growth rates, in percentages, of the quarterly real GDP of United Kingdom, Canada, and the United States used in the chapter. Fit a VAR(4) model to the series, simplify the model by...
-
Consider the U.S. quarterly gross private saving (GPSAVE) and gross private domestic investment (GPDI) from first quarter of 1947 to the third quarter of 2012. The data are from the Federal Reserve...

Study smarter with the SolutionInn App


