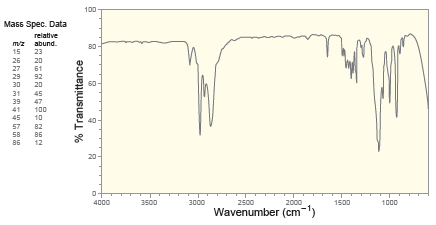
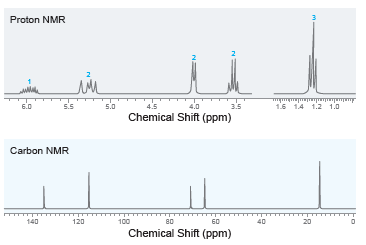
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the
Question:
Transcribed Image Text:
100 Mass Spec. Data relative mz abund. 15 23 26 27 29 20 61 92 30 20 31 39 47 41 100 45 10 57 82 58 86 86 12 20 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-") % Transmittance Proton NMR 1.5 14 1.2 1.0 6.0 5.5 5.0 4.5 4.0 3.5 Chemical Shift (ppm) Carbon NMR 140 120 100 80 60 20 Chemical Shift (ppm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Deduce the structure of a compound with molecular formula C 9 H 10 O 2 that produces the following 1 H NMR spectrum and 13 C NMR spectrum: Proton NMR 10 Chemical Shift (ppm) Carbon NMR - 128.4 128.8-...
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
The 1 H NMR spectrum of a compound with molecular formula C 7 H 15 C l exhibits two signals with relative integration 2 : 3. Propose a structure for this compound.
-
You have been given the following algorithm. What does it return? Input: Array A [ 1 . . . [ 1 . . . n ] ] Output: ? ? ? ? out 1 1 for i 2 2 to n do if A [ [ i ] < ] < A [ [ out ] ] then out i return...
-
What is cyber squatting?
-
Use a statistical spreadsheet or a graphing calculator with advanced statistical features to draw a scatter diagram of the data in Table 1. Then determine the linear correlation between club-head...
-
An engineering student correctly answers \(85 \%\) of all questions she attempts. What is the probability that the first incorrect answer was the fourth one?
-
Assume that you are purchasing an investment and have decided to invest in a company in the digital phone business. You have narrowed the choice to Best Digital Corp. and Every Zone, Inc. and have...
-
Lagoon Plc's Directors are planning to instigate the removal of its auditors, KPNG due to their inability to deliver "quality audit works". However, the Audit Manager has insisted that this move by...
-
Refer to Figure a, which shows a water tank subjected to a blast force f(t). We will model the tank and it's supporting columns as the mass-spring system shown in part (b) of the figure. The blast...
-
Deduce the structure of a compound with molecular formula C 8 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 2500 2000 Wavenumber (cm-1) 1000 4000 3500 3000...
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
In the normal distribution, 99.7% of the population lies within a. 1 standard deviation of the mean. b. 2 standard deviations of the mean. c. 3 standard deviations of the mean. d. 4 standard...
-
Hottie Inc., has the following standard and actual costs for the period. Hottie Inc Standard Costs Costs Standard Standard cost per unit Direct Materials 15 pounds @$4/pound 60 Direct Labor 3 hours...
-
Nicole decides that paying a $150 a month premium for car insurance is worth it because she will be sharing her liability with the insurance company this is an example of making a rational decision...
-
Describe the similarities about "The Art World's Amazing Spider Man", Mel Chin y "Wheatfields" de Agnes Denes?
-
Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise inventory Prepaid expenses Plant assets, net Total assets Liabilities and Equity...
-
Office Palace, Inc., signed a contract to lease an all-in-one printer to a new customer, Ashley, on December 27, 2023. The contract called for rent of $600 per month for a period of 36 months...
-
How does the recognition of revenue earned on account affect the income statement compared to its effect on the statement of cash flows?
-
Open Text Corporation provides a suite of business information software products. Exhibit 10-9 contains Note 10 from the companys 2013 annual report detailing long-term debt. Required: a. Open Text...
-
Draw and name the 17 hexene isomers, C6H2, including E, Z isomers.
-
Trans-2-Buterw is more stable than cic-2-hutene by only 4kJ/mol, but trans-2, 2, 5; 5-tetramethyl-3-hexene is more stable than its cis isomer by 3kJ/mol. Explain.
-
Cyclodecene can exist in both cis and Trans forms, but cyclohexene cannot. Explain. (Making molecular models is helpful.)
-
Assume that a robot systems constant failure and repair rates are 0.0004 failures per hour and 0.002 repairs per hour, respectively. Calculate the robot system steady-state unavailability and...
-
March 6, 2018 David Beckham's Latest Pitch: Men's Cosmetics. By Robert Williams Men of the world, David Beckham has a message for you: Moisturize. "It's the biggest thing," he says in a London...
-
3. On January 1, an investment fund was opened with an initial balance of 5000. Just after the balance grew to 5200 on July 1, an additional 2600 was deposited. The annual effective yield rate for...

Study smarter with the SolutionInn App


