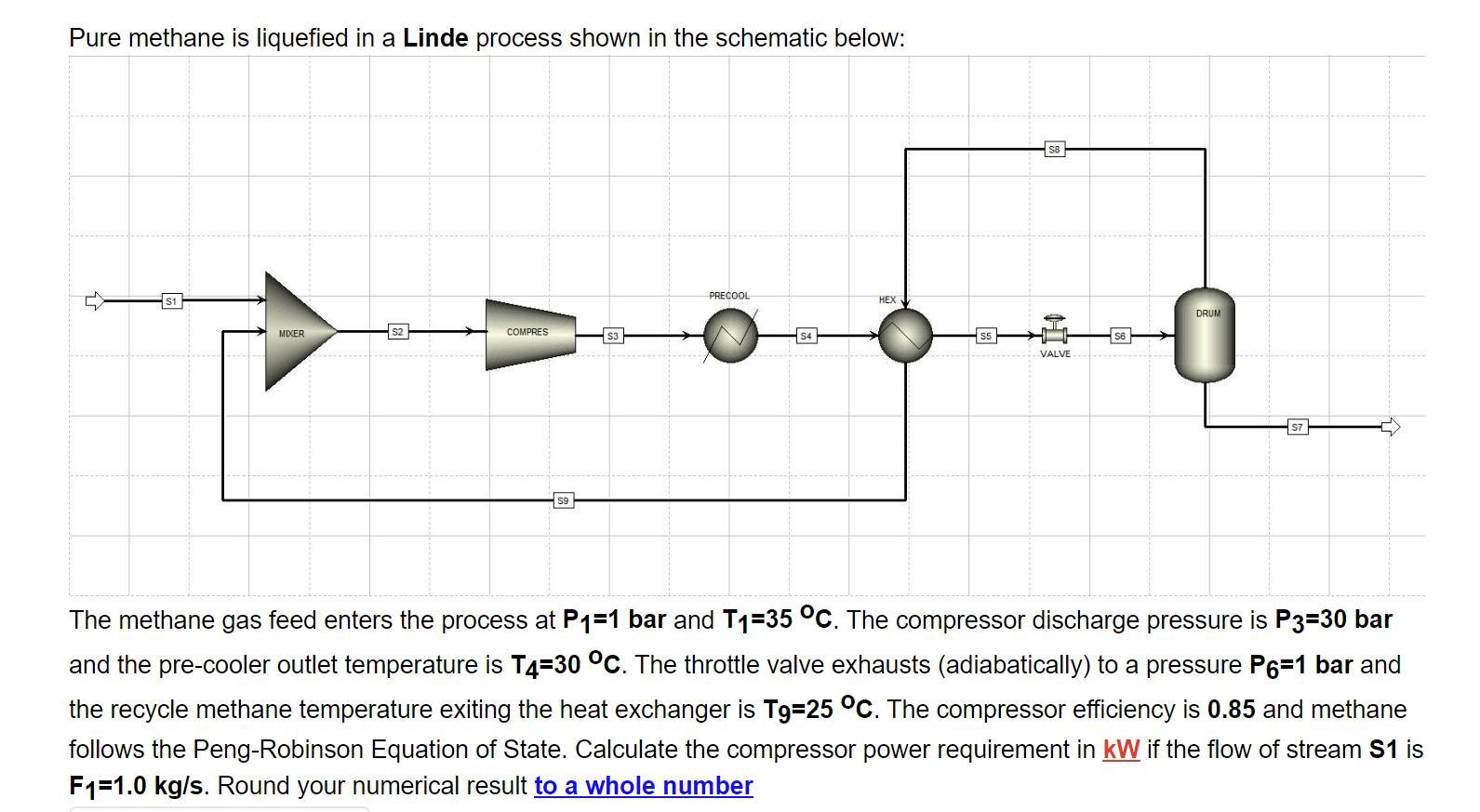
Pure methane is liquefied in a Linde process shown in the schematic below: S1 MIXER S2...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Pure methane is liquefied in a Linde process shown in the schematic below: S1 MIXER S2 COMPRES PRECOOL S4 HEX S5 VALVE 56 DRUM The methane gas feed enters the process at P=1 bar and T-35 C. The compressor discharge pressure is P3=30 bar and the pre-cooler outlet temperature is T4-30 C. The throttle valve exhausts (adiabatically) to a pressure P6=1 bar and the recycle methane temperature exiting the heat exchanger is T9=25 C. The compressor efficiency is 0.85 and methane follows the Peng-Robinson Equation of State. Calculate the compressor power requirement in kW if the flow of stream S1 is F1=1.0 kg/s. Round your numerical result to a whole number Pure methane is liquefied in a Linde process shown in the schematic below: S1 MIXER S2 COMPRES PRECOOL S4 HEX S5 VALVE 56 DRUM The methane gas feed enters the process at P=1 bar and T-35 C. The compressor discharge pressure is P3=30 bar and the pre-cooler outlet temperature is T4-30 C. The throttle valve exhausts (adiabatically) to a pressure P6=1 bar and the recycle methane temperature exiting the heat exchanger is T9=25 C. The compressor efficiency is 0.85 and methane follows the Peng-Robinson Equation of State. Calculate the compressor power requirement in kW if the flow of stream S1 is F1=1.0 kg/s. Round your numerical result to a whole number
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
At the head of a methane gas well in western Pennsylvania, the pressure is 250 bar, and the temperature is roughly 300 K. This gas stream is similar to the high-pressure stream exiting the precooler...
-
Make a thermodynamic analysis of the process described in Ex. 9.3. T = 295 K. Ex. 9.3 Natural gas, assumed here to be pure methane, is liquefied in a Claude process. Compression is to 60 bar and...
-
According to a study by the American Pet Food Dealers Association, 63% of U.S. house-holds own pets. A report is being prepared for an editorial in the San Francisco Chronicle. As a part of the...
-
Find the direction and magnitude of the vector 2 A(vector) + B(vector), where A(vector) = (12.1 m) x and B(vector) = (32.2 m)y.
-
A comparative balance sheet for Shabbona Corporation is presented as follows. Additional information: 1. Net income for 2020 was $125,000. No gains or losses were recorded in 2020. 2. Cash dividends...
-
A representative volume element (RVE) consisting of a cylindrical isotropic fiber embedded and perfectly bonded in a cylinder of isotropic matrix material is shown in Figure 5.23. If the ends of the...
-
The general ledger of Red Storm Cleaners at January 1, 2021, includes the following account balances: The following is a summary of the transactions for the year:1. March 12 Provide services to...
-
In a criminal trial, the prosecution seeks to prove that the defendant, Calvin Broadus robbed a store. There was a witness named Christopher Wallace who told a police officer named Jermaine Cole that...
-
Compte, Incorporated (a U.S.-based company), establishes a subsidiary in Croatia on January 1, 2023. The following account balances for the year ending December 31, 2024, are stated in kuna (K), the...
-
Kaizen represents the method for using cross-functional teams to drive improvement initiatives. Explain the key factors for practical Kaizen training including communication, grasping the situation,...
-
In below picture, the Temperature-Time-Transition (TTT) diagram for a glass forming material is shown: temperature T. m I 1 ppm 1% crystal time
-
1. Zachary Fox does not make any voluntary deductions that impact eamings subject to federal income tax withholding or FICA taxes. He has authorized voluntary deductions each pay period of $10 for...
-
Amazon Sales $177,866 Accounts receivable: Beginning of year 20,816 End of year 16,677 Determine the accounts receivable turnover. For interim computations, carry amounts out to one decimal place....
-
2. If a person seeks to report fraud related to a World Bank Group financed operation, to what department within the World Bank should that person direct their report? Describe the process of how the...
-
When a company lends money to employees at a rate of 4%, the company will record ______. Multiple choice question. a liability called Accounts Payable a liability called Notes Payable an asset called...
-
Identify and describe an environmental issue and a proposed way to reduce impact and improve outcomes. The proposal could be as simple as a campaign to convince people to change daily habits that are...
-
Annual dividends of ATTA Corp grew from $0.96 in 2005 to $1.76 in 2017. What was the annual growth rate?
-
In Exercises find all points (if any) of horizontal and vertical tangency to the curve. Use a graphing utility to confirm your results. x = 2 + 2 sin , y = 1 + cos
-
In Exercises sketch the region whose area is given by the definite integral. Then use a geometric formula to evaluate the integral (a > 0, r > 0). *7 [ -7 49 - x dx
-
In Exercises match the sequence with its graph. [The graphs are labeled (a), (b), (c), and (d).] (a) (b) (c) (d) 9 an 5 4 3 2- I 2 u ||||||| 4 6 8 10
-
With reference to Example 2, Chapter 2, test that the mean copper content is the same for both heats. Data From Example 2 Data From Figure 3.2 EXAMPLE 2 Relation of regions in Venn diagrams to events...
-
With reference to Example 8, find a \(90 \%\) confidence interval for the difference of mean strengths of the alloys (a) using the pooled procedure; (b) using the large samples procedure. Data From...
-
With reference to part (a) of Exercise 8.33, how would you pair and then randomize for a paired test? Data From Exercise 8.33 8.33 How would you randomize, for a two sample test, in each of the...

Study smarter with the SolutionInn App


