8) Answer question 1 from Chapter 4 in your book. You may use excel to answer...
Fantastic news! We've Found the answer you've been seeking!
Question:

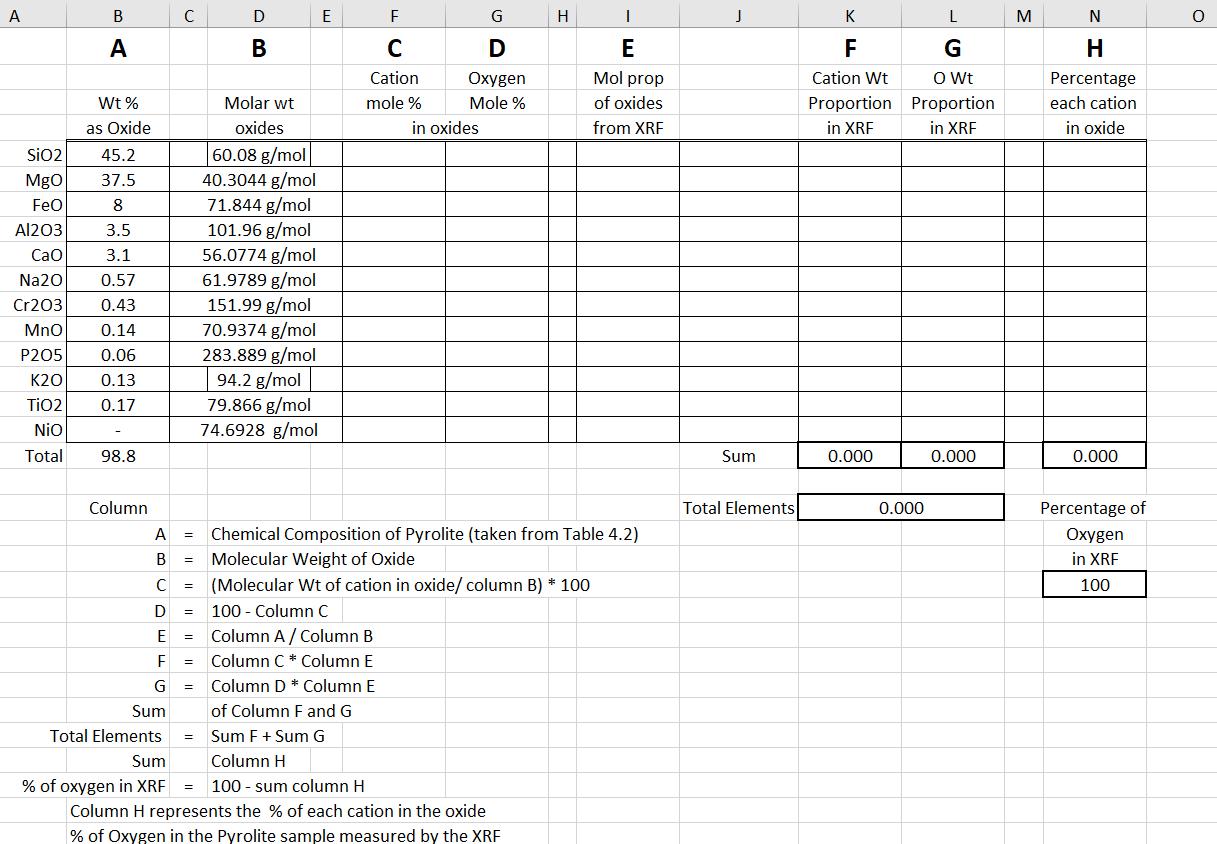
Transcribed Image Text:
8) Answer question 1 from Chapter 4 in your book. You may use excel to answer this question. Just make sure to provide an example of your calculations with it. Hints: a. Determine the molar weight of each oxide b. Determine the % of each cation and oxygen in the oxide. This will be the cation and oxygen mole% c. Determine the ratio of the weight % of the oxide to its molar weight. This is useful converting the elemental concentration as molar to weight. d. Multiply the ratio from part c by each cation mole% to get the weight of each cation in the oxide. e. Multiply the ratio from part c by each oxygen mole% to get the weight of each oxygen in the oxide. f. Sum the weights of each cation to get a total cation weight. g. Sum the weights of each oxygen to get a total oxygen weight. h. Sum the total cation weight with the total oxygen weight to get the total elemental weight in the sample. i. Divide each cation weight from part e by the total elemental weight to get a weight % for each cation in the sample j. Divide the total oxygen weight from part g by the total elemental weight to get the weight% of oxygen in the sample. A B C A A В DB Wt% as Oxide Molar wt oxides 60.08 g/mol J K L M N F from XRF Cation Wt Proportion in XRF G O Wt Proportion in XRF each cation in oxide H Percentage E F G H C Cation mole % D Oxygen Mole % Mol prop of oxides E E in oxides SiO2 45.2 MgO 37.5 40.3044 g/mol FeO 8 71.844 g/mol Al203 3.5 101.96 g/mol CaO 3.1 56.0774 g/mol Na20 0.57 61.9789 g/mol Cr203 0.43 151.99 g/mol MnO 0.14 70.9374 g/mol P205 0.06 283.889 g/mol K20 0.13 94.2 g/mol TiO2 0.17 79.866 g/mol NiO 74.6928 g/mol Total 98.8 Column Sum 0.000 0.000 0.000 A B = Chemical Composition of Pyrolite (taken from Table 4.2) Molecular Weight of Oxide (Molecular Wt of cation in oxide/ column B) * 100 Total Elements C = D 100 - Column C E = Column A/Column B F = G Sum Total Elements Sum Column C * Column E Column D * Column E of Column F and G = Sum F + Sum G Column H % of oxygen in XRF = 100 sum column H Column H represents the % of each cation in the oxide % of Oxygen in the Pyrolite sample measured by the XRF 0.000 Percentage of Oxygen in XRF 100 8) Answer question 1 from Chapter 4 in your book. You may use excel to answer this question. Just make sure to provide an example of your calculations with it. Hints: a. Determine the molar weight of each oxide b. Determine the % of each cation and oxygen in the oxide. This will be the cation and oxygen mole% c. Determine the ratio of the weight % of the oxide to its molar weight. This is useful converting the elemental concentration as molar to weight. d. Multiply the ratio from part c by each cation mole% to get the weight of each cation in the oxide. e. Multiply the ratio from part c by each oxygen mole% to get the weight of each oxygen in the oxide. f. Sum the weights of each cation to get a total cation weight. g. Sum the weights of each oxygen to get a total oxygen weight. h. Sum the total cation weight with the total oxygen weight to get the total elemental weight in the sample. i. Divide each cation weight from part e by the total elemental weight to get a weight % for each cation in the sample j. Divide the total oxygen weight from part g by the total elemental weight to get the weight% of oxygen in the sample. A B C A A В DB Wt% as Oxide Molar wt oxides 60.08 g/mol J K L M N F from XRF Cation Wt Proportion in XRF G O Wt Proportion in XRF each cation in oxide H Percentage E F G H C Cation mole % D Oxygen Mole % Mol prop of oxides E E in oxides SiO2 45.2 MgO 37.5 40.3044 g/mol FeO 8 71.844 g/mol Al203 3.5 101.96 g/mol CaO 3.1 56.0774 g/mol Na20 0.57 61.9789 g/mol Cr203 0.43 151.99 g/mol MnO 0.14 70.9374 g/mol P205 0.06 283.889 g/mol K20 0.13 94.2 g/mol TiO2 0.17 79.866 g/mol NiO 74.6928 g/mol Total 98.8 Column Sum 0.000 0.000 0.000 A B = Chemical Composition of Pyrolite (taken from Table 4.2) Molecular Weight of Oxide (Molecular Wt of cation in oxide/ column B) * 100 Total Elements C = D 100 - Column C E = Column A/Column B F = G Sum Total Elements Sum Column C * Column E Column D * Column E of Column F and G = Sum F + Sum G Column H % of oxygen in XRF = 100 sum column H Column H represents the % of each cation in the oxide % of Oxygen in the Pyrolite sample measured by the XRF 0.000 Percentage of Oxygen in XRF 100
Expert Answer:
Answer rating: 100% (QA)
SOLUTION To answer this question we need to use the information given in the table above which shows the chemical composition of pyrolite taken from Table 42 and the molecular weight of each oxide Ste... View the full answer

Posted Date:
Students also viewed these accounting questions
-
Entry for Jobs Completed; Cost of Unfinished Jobs The following account appears in the ledger prior to recognizing the jobs completed in August: Work in Process $9,100 73,980 79,810 41,950 Balance,...
-
Homework In a location of highway construction project, where the (cross and longitudinal ) sections of the earthworks profile in that location are as shown in the given figure. Estimate the totally...
-
Homework of Earthworks In a location of highway construction project , where the ( cross and longitudinal ) sections of the earthworks profile in that location are as shown in the given figure....
-
The table contains real data. MySpace U.S. Advertising Revenue ($ millions) (a) Determine the maximum and minimum values for each variable in the table. (b) Use your results from part (a) to find an...
-
A generation ago men were more likely to attend university and acquire a graduate degree than women. However, women now appear to be attending university in greater numbers than men. To gauge the...
-
Two producers having equal positions on a market offer two substitute prod- ucts. The demand for these products evolves according to the following demand functions: y (P, P2) = ap +2 +, == y (P1,...
-
Use a stem-and-leaf plot to display the data, which represent the runs scored by a batsman in a World Cup series. Organize the data using the indicated type of graph. Describe any patterns. 70 75 71...
-
Model X100 sells for $120 per unit whereas Model X200 offers advanced features and sells for S500 per unit. Management expects to sell 50,000 units of Model X100 and 5,000 units of Model X200 next...
-
Ethics in Human Resource Practices HR teams will play vital roles in organizations to ensure ethical behavior and compliance. To be effective, they must first place an emphasis on ethical behavior...
-
A developer has a rectangular 5-acre site with a commercial zoning designation allowing mini-storage, office, retail, or apartments. The land is under contract for purchase for $3.35 million and the...
-
13. Write the output of the following code for i in range(0,5): if i%2=0: for j in range(0,5): if j%2=0: print(1,end='') elif j%2!=0: print(0,end='') print(") elif i%2!=0: for j in range(0,5): if...
-
Fiscal Policy responses of European countries in fighting the great recession in the aftermath of the financial crisis and the European sovereign debt crisis in the years after 2009 differed across...
-
A branch manager realized that 2 of his former employees had deceived some customers. Although no customer has approached us so far, the branch manager finds himself in an ethical dilemma to report...
-
The symphony ticket market can be described by the following supply and demand curves: Q D = 50.000 100P y Q S = 20.000 + 100P What are the equilibrium price and quantity in the ticket market?...
-
Calculate PPP-adjusted GDP for each of the four countries, using the information in the table below. Instructions: Round your answers to the nearest dollar. Country PIB ps Price level (%) GDP...
-
If the marginal cost of the 1,000th unit produced by a monopolist is $16 and its marginal revenue is $20, what should the monopolist do?
-
Table below indicates the duration of and labour requirement for each activity of a small project. Duration (Weeks) Gang Size 10 Activity 1-2 2 2-3 2-4 2 3 2 3-5 4 4-5 8 563 4-6 4 4 5-6 1 2 5-7 8 8...
-
In Exercises 15 through 30, find the derivative dy/dx. In some of these problems, you may need to use implicit differentiation or logarithmic differentiation. y ex + et -2x 1 + e
-
The following is the distribution of the hourly number of trucks arriving at a company's warehouse: Find the mean of this distribution, and using it (rounded to one decimal place) as the parameter...
-
With reference to Exercise 10.42, verify that the mean of the observed distribution is 1.6 , corresponding to \(40 \%\) of the cars requiring repairs. Then look up the probabilities for \(n=5\) and...
-
Among 100 purification filters used in an experiment, 46 had a service life of less than 20 hours, 19 had a service life of 20 or more but less than 40 hours, 17 had a service life of 40 or more but...

Study smarter with the SolutionInn App

