Q5: A quantity of 1.4 moles of an ideal gas go through the 3 processes described...
Fantastic news! We've Found the answer you've been seeking!
Question:
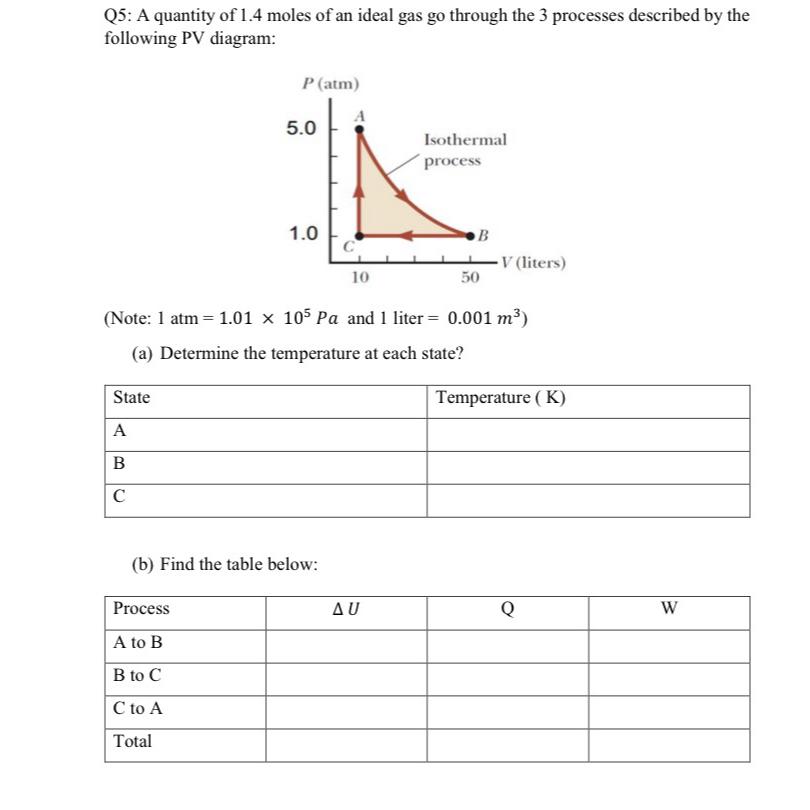
Transcribed Image Text:
Q5: A quantity of 1.4 moles of an ideal gas go through the 3 processes described by the following PV diagram: State A B P (atm) 5.0 Process A to B B to C C to A Total 1.0 (b) Find the table below: C 10 Isothermal process (Note: 1 atm = 1.01 105 Pa and 1 liter = 0.001 m) (a) Determine the temperature at each state? AU B 50 V (liters) Temperature (K) Q W Q5: A quantity of 1.4 moles of an ideal gas go through the 3 processes described by the following PV diagram: State A B P (atm) 5.0 Process A to B B to C C to A Total 1.0 (b) Find the table below: C 10 Isothermal process (Note: 1 atm = 1.01 105 Pa and 1 liter = 0.001 m) (a) Determine the temperature at each state? AU B 50 V (liters) Temperature (K) Q W
Expert Answer:
Answer rating: 100% (QA)
For an ideal gas the state equation PV nRT can be used where P is pressure V is volume n is the number of moles R is the ideal gas constant and T is t... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
Consider now the dynamic power dissipation of different versions of a given processor for three different voltages given in the following table. Determine the static and dynamic power dissipation...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
You are the president of a new, capital-rich firm and you must decide on your firm's targeting approach. Three large subgroups exist in your market that have never had their unique needs served. If...
-
The following information is for a firm that has net debt on its balance sheet (in millions of dollars). Common shareholders' equity, December 3 1, 2011 ...................... 174.8 Common dividends,...
-
In Exercises determine whether the statement is true or false. If it is false, explain why or give an example that shows it is false. If D 0 or E 0, then the graph of y - x + Dx + Ey = 0 is a...
-
A university pumps its water from wells located on campus. The falling water table has caused pumping costs to increase, the quantity of water available to decrease, and the quality of water to...
-
For the year ended December 31, 2014, Denkinger Electrical Repair Company reports the following summary payroll data. Gross earnings: Administrative salaries ............$200,000 Electricians wages...
-
Data Manipulation Tasks a Insert 20 records into each table for testing purposes. a Delete an entire order by using the unique identifier for that order. o Update the price of a product by using the...
-
Suppose that Kate and Anne enter into a pooling arrangement. Assume that both women have the following loss distributions and that losses are independent. $50,000 with probability of 0.005 $20,000...
-
Define the following? Data Message Datum
-
For both Twitter and Facebook forecasts the Income Statement Uses the sales growth assumption to forecast sales for the next fiscal year. Use costlevel assumptions to forecast all the operating...
-
Suppose that a stock price has an expected return of 16% per annum and a volatility of 30% per annum. When the stock price at the end of a certain day is $50, calculate the following: (a) The...
-
Companys dividend next year is expected to be $1.00 (D1). The stock price is $20. The company is expected to have a constant 4.25% constant growth rate in dividends. What is the companys cost of...
-
Is the answer correct? The graph of a y = f(x) is shown. 6 + 5 14 m ~ 1 -6 -5 -4 -3 2 -1 -1 -2 -3 A w -4 -5- -6+ 1 2 3 4 The domain of f(x) is x = [-3,3] The domain of g(x) = notation) S 6 f(x) is a ...
-
Let f(x) = 1 x-4 g(f(x)) = and g(x)= = 3 - X +4. Find the following functions. Simplify your answers. f(g(x)) =
-
x*+2y* Evaluate // dV, where D is the region enclosed by the spherical surface D 2x4+8y++2z+ x2 + y? + z? = 1. Correct the result to 4 decimal places.
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
The irreversible gas-phase reaction Acat B is carried out adiabatically over a packed bed of solid catalyst particles. The reaction is first order in the concentration of A on the catalyst surface...
-
Read the Web Module Chemical Reaction Engineering of Hippopotamus Stomach on the CRE Web site (http://www.umich.edu/~elements/6e/web_mod/hippo/index.htm). a. Work problems (1) and (2) in the Hippo...
-
Go to the Web site (http://www.umich.edu/~elements/6e/06chap/iclicker_ch6_q1.html) and view at least five i>clicker questions. Choose one that could be used as is, or a variation thereof, to be...
-
Classify each of the following as either a policy instrument or an intermediary target. Explain your answer. a. Long-term interest rates b. Central bank interest rates c. M2 d. Reserve requirements
-
The New York Fed executes foreign exchange interventions for the Federal Reserve system. Go to https://www.newyorkfed.org/markets/quar_reports.html to see quarterly summaries of the Feds foreign...
-
The New Zealand dollar to U.S. dollar exchange rate is 1.38, and the British pound to U.S. dollar exchange rate is 0.65. If you find that the British pound to New Zealand dollar is trading at 0.5,...

Study smarter with the SolutionInn App


