Question: Question 1 (15 points): Draw the structure of ethyl cyclohexanecarboxylate in the box below and provide the major products of each of the following
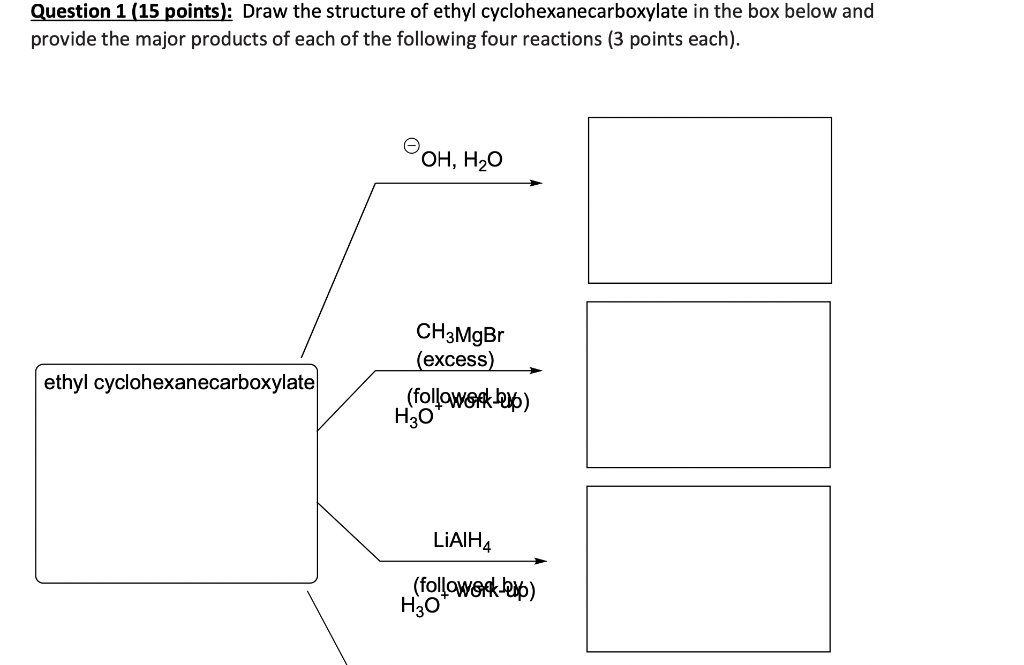
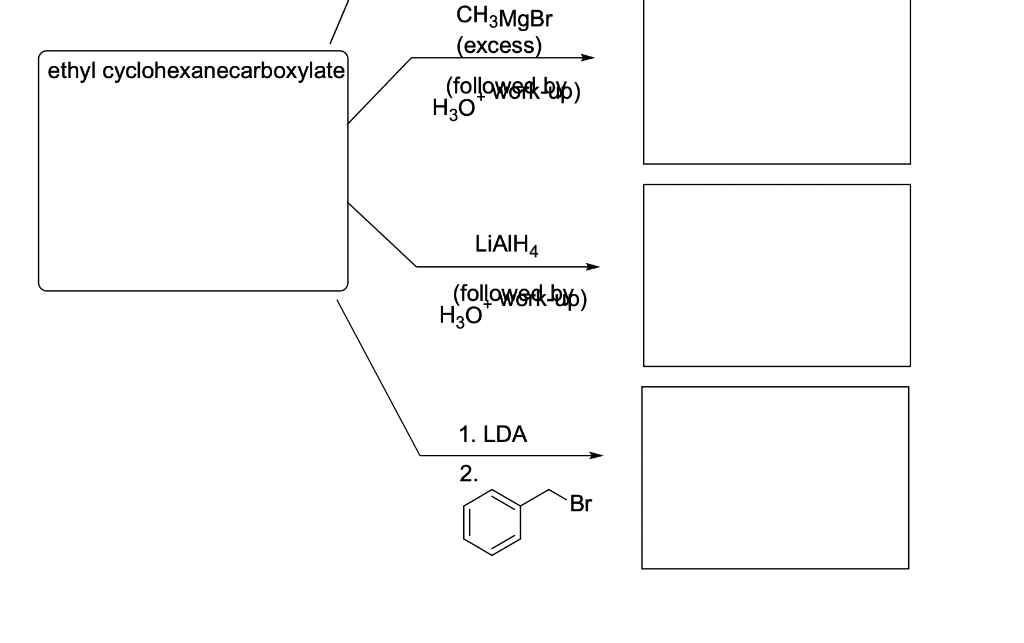
Question 1 (15 points): Draw the structure of ethyl cyclohexanecarboxylate in the box below and provide the major products of each of the following four reactions (3 points each). ethyl cyclohexanecarboxylate OH, HO CH3MgBr (excess) (followork) HO LIAIH4 (followork-up) H3O ethyl cyclohexanecarboxylate CH 3MgBr (excess) (followork up) H3O LIAIH4 (followerck-up) H3O 1. LDA 2. Br
Step by Step Solution
There are 3 Steps involved in it
1 Ester on reaction with OH and H 2 O undergo base hydrolysis and yield carboxylic acid 2 E... View full answer

Get step-by-step solutions from verified subject matter experts


