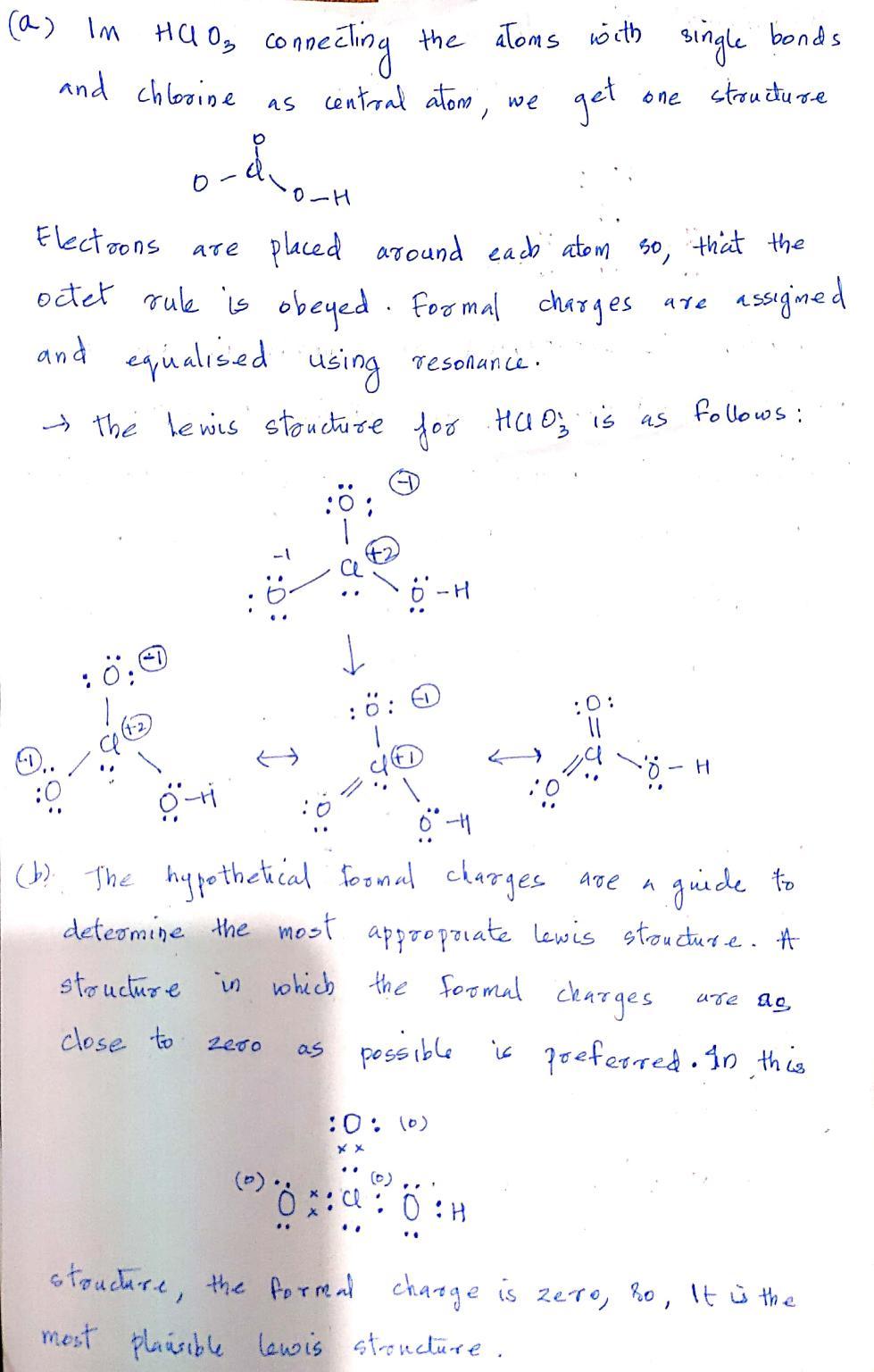
(a) Im HClO connecting the atoms with single bonds and chlorine as central atom, one structure...
Fantastic news! We've Found the answer you've been seeking!
Question:
![3 Chlorine is surrounded by pair of electrons, so, the steric E a Centroal atom] X O (d) In HOO, CH With](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/03/623415dd09149_636623415dc94f6a.jpg)
Transcribed Image Text:
(a) Im HClO₂ connecting the atoms with single bonds and chlorine as central atom, one structure we get D 1.. :0 -&-o-H он Electrons octet rule is obeyed. Formal charges using resonance. and equalised using → the lewis structure for HCO3 is as follows: are placed around each atom so, that the assigned 2000 -H :0 (0) xx O-H (h) The hypothetical formal charges are a guide to determine the most appropriate lewis structure. A in which the formal charges structure are ag close to as possible is preferred. In this are *:: 0 :H H structure, the formal charge is zero, ho, It is the most plausible lewis structure. 3₁ Chlorine is surrounded by pair of electrons, so, the steric E a Centroal atom] X→ O (d) In HOO₂, CH → With steric no 4. it has one pair of lone pair. so its molecular geometry is Trigonal pyramidal → For Trigonal pyramidal shaped molecules the appross- mate Bond angle is lege. has colorine The hybridisation of U is sp³. [Ne] 38² 3p5. 35 LS 3 bonds and [12] [72/12/12] 25 2P undergone sp² hybridisation. The electron configuration for chlorine ic. 11 35 one Sone i4. XX Trigonal pyramidal 1090. is present in ground state. 11 11 7 3.p (a) Im HClO₂ connecting the atoms with single bonds and chlorine as central atom, one structure we get D 1.. :0 -&-o-H он Electrons octet rule is obeyed. Formal charges using resonance. and equalised using → the lewis structure for HCO3 is as follows: are placed around each atom so, that the assigned 2000 -H :0 (0) xx O-H (h) The hypothetical formal charges are a guide to determine the most appropriate lewis structure. A in which the formal charges structure are ag close to as possible is preferred. In this are *:: 0 :H H structure, the formal charge is zero, ho, It is the most plausible lewis structure. 3₁ Chlorine is surrounded by pair of electrons, so, the steric E a Centroal atom] X→ O (d) In HOO₂, CH → With steric no 4. it has one pair of lone pair. so its molecular geometry is Trigonal pyramidal → For Trigonal pyramidal shaped molecules the appross- mate Bond angle is lege. has colorine The hybridisation of U is sp³. [Ne] 38² 3p5. 35 LS 3 bonds and [12] [72/12/12] 25 2P undergone sp² hybridisation. The electron configuration for chlorine ic. 11 35 one Sone i4. XX Trigonal pyramidal 1090. is present in ground state. 11 11 7 3.p
Expert Answer:
Answer rating: 100% (QA)
It seems like youre discussing the molecular geometry and hybridization of chlori... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
Le Gourmand is one of Canadas most famous French restaurants. Located 20 kilometers north of Toronto, it draws diners from all over the province to enjoy its fare. One of the attractions at the...
-
A Lewis structure obeying the octet rule can be drawn for O2 as follows: Use the molecular orbital energy- level diagram for O2 to show that the above Lewis structure corresponds to an exicted state....
-
The octet rule correctly predicts the Lewis formula of many molecules involving main group elements. Explain why this is so.
-
Why does the following code fragment not have the same effect as the code fragment in the previous question? x.next t.next = t; = x.next;
-
Laura recently was diagnosed with cancer and has begun chemotherapy treatments. A cancer specialist has given Laura less than one year to live. She has incurred sizable medical bills and other...
-
Marvel Media, LLC, has three members: WLKT Partners, Madison Sanders, and Observer Newspaper, LLC. On January 1, 2016, the three members had equity of $200,000, $40,000, and $160,000, respectively....
-
The concentration of $\mathrm{NbOCl}_{3}$ in a tube-flow reactor as a function of several controllable variables is shown in Table B.6. a. Fit a multiple regression model relating concentration of...
-
Consider a gas-fired boiler in which five coiled thin-walled, copper tubes of 25-mm diameter and 8-m length are submerged in pressurized water at 4.37 bars. The walls of the tubes are polished and...
-
There are only 4 teams in a league. The winning percentages of the teams are given as follows: Team A: 58%, Team B: 30%, Team C: 62% and Team D: 82%. Calculate the standard deviation.
-
If the Sommers had chosen the original 15-year, 6.25% mortgage proposal, how much tax shelter would they have lost (over the last five years) as compared to the 30-year, 7.25% mortgage?
-
The plastic surgery department has annual costs of $18m and annual charges of $36m. The department charges $720 for mole removal. According to the RCC approach, what does it cost the department to...
-
Which is the definitio Process of partcipating firsthand in the communication process as a listener, rather than just passively listening to what is said. Process of hearing what others say with the...
-
The associated perceptual mapping software, describe the two ( or , if applicable, three ) dimensions underlying the perceptual maps that you generated. Based on these maps, how do people in the...
-
For a sport facility to be effective and achieve its goals and objectives, the ownership and the governance structures must be able to successfully respond to changes in its environment.Changes in...
-
How does that help you understand why a person might not be a hypocrite even if they behave in a way that appearss different from their attitude?
-
What roles have social factors ( beyond the sphere of health care orpublic health ) played in controlling or failing to control the HIV / AIDS epidemic?
-
Assignment - Lift Controller 1. Aims To work in a group to produce a digital design. To design a simple system using VHDL. To use a modular approach to design a simple LIFT CONTROLLER. To simulate...
-
During the year land was revalued and the surplus reported as Revaluation surplus; and an asset costing 80,000, written down to 38,000, was sold for 40,000. Identify the cost of any non-current...
-
Calculate the percentage by mass of oxygen in the following compounds: (a) Morphine, C17H19NO3 (b) Codeine, C18H21NO3 (c) Cocaine, C17H21NO4 (d) Tetracycline, C22H24N2O8 (e) Digitoxin, C41H64O13 (f)...
-
In the accompanying cylinder diagram a chemical process occurs at constant temperature and pressure. (a) Is the sign of w indicated by this change positive or negative? (b) If the process is...
-
Identify the specific element that corresponds to each of the following electron configurations and indicate the number of unpaired electrons for each: (a) 1s 2 2s 2 , (b) 1s 2 2s 2 2p 4 , (c) [A]4s...
-
Companies are facing a great amount of change in every facet of their operations today. To remain competitive, companies must keep abreast of current developments in several areas. You recently got...
-
For each of the situations listed, identify the primary standard from the IMA Statement of Ethical Professional Practice that is violated (competence, confidentiality, integrity, or credibility.)...
-
Complete the following statements with one of the terms listed here. You may use a term more than once, and some terms may not be used at all. a. To quickly navigate to the end of the data in a...

Study smarter with the SolutionInn App


