QUESTION 13 An atom of carbon has six protons. What will happen if the number of...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
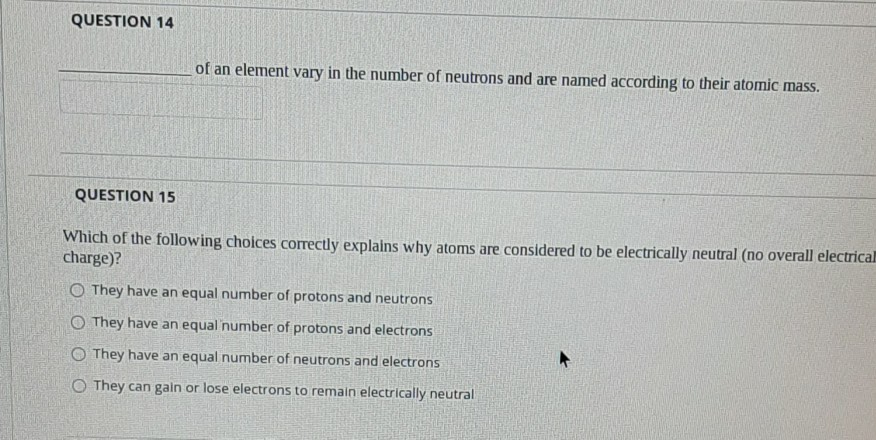
QUESTION 13 An atom of carbon has six protons. What will happen if the number of protons is increased to 7? Would it still be carbon? Refer to the periodic table below if needed. Periodic Table of the Elements Os Ba Tv MA 44 2228 44 ERE PALE Oa. Yes, it will still be carbon but it will be a heavier Isotope Ob. It will no longer be carbon, because the number of protons determines the specific element. Oc. It will no longer be carbon, an element with an atomic number of 7 is Nitrogen. O d. Yes, it will be carbon but it would be an ion with a positive charge. e. Both b and care true. QUESTION 14 QUESTION 15 of an element vary in the number of neutrons and are named according to their atomic mass. Which of the following choices correctly explains why atoms are considered to be electrically neutral (no overall electrical charge)? O They have an equal number of protons and neutrons O They have an equal number of protons and electrons They have an equal number of neutrons and electrons O They can gain or lose electrons to remain electrically neutral QUESTION 13 An atom of carbon has six protons. What will happen if the number of protons is increased to 7? Would it still be carbon? Refer to the periodic table below if needed. Periodic Table of the Elements Os Ba Tv MA 44 2228 44 ERE PALE Oa. Yes, it will still be carbon but it will be a heavier Isotope Ob. It will no longer be carbon, because the number of protons determines the specific element. Oc. It will no longer be carbon, an element with an atomic number of 7 is Nitrogen. O d. Yes, it will be carbon but it would be an ion with a positive charge. e. Both b and care true. QUESTION 14 QUESTION 15 of an element vary in the number of neutrons and are named according to their atomic mass. Which of the following choices correctly explains why atoms are considered to be electrically neutral (no overall electrical charge)? O They have an equal number of protons and neutrons O They have an equal number of protons and electrons They have an equal number of neutrons and electrons O They can gain or lose electrons to remain electrically neutral
Expert Answer:
Answer rating: 100% (QA)
13 e Both b and c are true The carbon atom no longer remain same and becomes ... View the full answer

Related Book For 

Foundations of Financial Management
ISBN: 978-1259024979
10th Canadian edition
Authors: Stanley Block, Geoffrey Hirt, Bartley Danielsen, Doug Short, Michael Perretta
Posted Date:
Students also viewed these biology questions
-
In the apparatus shown below, what will happen if the membrane is (a) Permeable to both water and the Na+ and Cl- ions, (b) Permeable to water and Na+ ions but not to Cl- ions, (c) Permeable to water...
-
What will happen if the Gram-Schmidt process is applied to a set of vectors {v2, v2, v3}, where v1 and v2 are Jinearly independent, but v3 Span(v1 v2). Will the process fail? If so how? Explain.
-
What do you predict will happen if the central bank takes no monetary policy actions? What monetary policy action would you advise the central bank to take and what do you predict will be the effect...
-
Explain factors governing recruitment?
-
Give two examples of events that could shift the demand for labor, and explain why they do so.
-
The Browns want to purchase a house that costs $150,000. They plan to take out a $120,000 mortgage on the house and put $30,000 as a down payment. The bank informs them that with a 15-year mortgage...
-
Locate questions that can be used during a debtors examination once a judgment is obtained. Try to find questions specific to California, as many states have limits on the types of questions that can...
-
On October 1, 2012, Holmgren Chemical was identified as a potentially responsible party by the Environmental Protection Agency. Holmgrens management along with its counsel have concluded that it is...
-
A woman invests $6400 in an account that pays 6% interest per year, compounded continuously. (a) What is the amount after 2 years? (Round your answer to the nearest cent.) $ 7215.98 (b) How long will...
-
One criticism of Hofstedes work is that the scores on each dimension reflect only an average tendency of a particular country and, therefore, inadequately reflect the wide range of responses given...
-
Record closing journal entries for the month ending January 2021. Use the Chart of Accounts provided below and be sure to enter the account titles exactly as they appear in the Chart of Accounts. For...
-
A customer has a concern of slow cranking only on a hot restart. During the visual inspection, vou also notice the insulation on the negative cable is miched. What may cause this problem? a. The...
-
In the image below the national brand is shown in the top left, and three store brand equivalents are shown in the other three corners. 1. ) Given the standard described in the Conopco case, which...
-
In the supplementary reading "When Do We Know What We Think We Know," http://www.econ.ucla.edu/people/papers/currie/more/ACH.pdf 1: what is the author Currie's perspective on whether evaluations...
-
1. Look for as many theories and concepts from your textbook (Leadership in the Global Context) that are relevant to a phase of your Life Design Program and Ledership Develop Program. 2. Draw as many...
-
In a negative let-off motion, the warp tension should be maintained at 600 N throughout the weaving process. The tight side tension of the chain is four times that of the slack side. The weight...
-
In which of these situations does it make sense to say economics predicts people will not internalize an externality, like pollution? (1 point) A. When the transaction costs of trading in rights to...
-
Solve the relation Exz:Solve therelation ne %3D
-
The following companies have different financial statistics. What dividend policies would you recommend for them? Explain your reasons. Turtle Co. Hare Corp. Growth rate in sales and earnings... Cash...
-
Compare the balance based on IFRS with that based on ASPE, and describe limitations of the balance sheet information for financial analysis.
-
The Thorpe Corporation will purchase a $50,000 piece of production machinery with an estimated useful life of five years. The new machine is expected to allow an increase in sales of $80,000 per...
-
The flow about a \(150 \mathrm{~mm}\) artillery projectile which travels at \(600 \mathrm{~m} / \mathrm{s}\) through still air at \(30^{\circ} \mathrm{C}\) and absolute pressure \(101.4...
-
Consider water flow around a circular cylinder, of diameter \(D\) and length \(l\). In addition to geometry, the drag force is known to depend on liquid speed, \(V\), density, \(ho\), and viscosity,...
-
A 1:10 scale model of a tractor-trailer rig is tested in a wind tunnel. The model frontal area is \(A_{m}=0.1 \mathrm{~m}^{2}\). When tested at \(V_{m}=75 \mathrm{~m} / \mathrm{s}\) in standard air,...

Study smarter with the SolutionInn App


