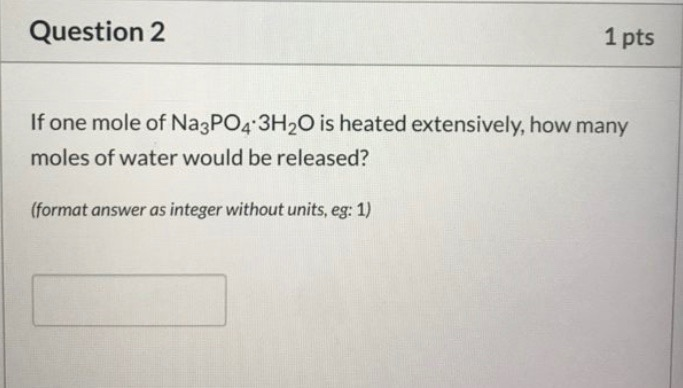
Question 2 1 pts If one mole of Na3PO4 3H2O is heated extensively, how many moles...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
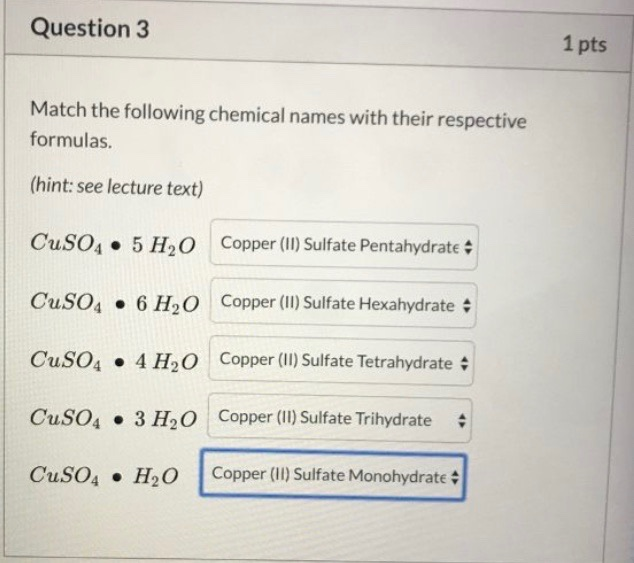
Question 2 1 pts If one mole of Na3PO4 3H2O is heated extensively, how many moles of water would be released? (format answer as integer without units, eg: 1) Question 3 Match the following chemical names with their respective formulas. (hint: see lecture text) CuSO4 5 H₂O CuSO4 6 H₂O CuSO4 4 H₂O CuSO4 3 H₂O CuSO4 H₂O Copper (II) Sulfate Pentahydrate # Copper (II) Sulfate Hexahydrate Copper (II) Sulfate Tetrahydrate Copper (II) Sulfate Trihydrate Copper (II) Sulfate Monohydrate # 1 pts Question 5 1 pts If a sample of a hydrate contains 0.02mol of anhydrous salt Cu (SO4)y and 0.1mol of water, how many water molecules are present in one formula unit of the hydrate (ie. what is z in the formula Cur (SO4)yzH₂O)? Enter your answer as an integer without units (eg. 2). Question 2 1 pts If one mole of Na3PO4 3H2O is heated extensively, how many moles of water would be released? (format answer as integer without units, eg: 1) Question 3 Match the following chemical names with their respective formulas. (hint: see lecture text) CuSO4 5 H₂O CuSO4 6 H₂O CuSO4 4 H₂O CuSO4 3 H₂O CuSO4 H₂O Copper (II) Sulfate Pentahydrate # Copper (II) Sulfate Hexahydrate Copper (II) Sulfate Tetrahydrate Copper (II) Sulfate Trihydrate Copper (II) Sulfate Monohydrate # 1 pts Question 5 1 pts If a sample of a hydrate contains 0.02mol of anhydrous salt Cu (SO4)y and 0.1mol of water, how many water molecules are present in one formula unit of the hydrate (ie. what is z in the formula Cur (SO4)yzH₂O)? Enter your answer as an integer without units (eg. 2).
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How many moles of water are there in 1.000 L? How many molecules?
-
If one mole of the following compounds were each placed into separate beakers containing the same amount of water, rank the Cl(aq) concentrations from highest to lowest (some may be equivalent): KCl,...
-
What different types of RNA molecules are present in prokaryotic cells? in eukaryotic cells? What roles do these different classes of RNA molecules play in the cell?
-
The adjusted trial balance for Sweet Home Catering, Inc., is presented below. Prepare the income statement and statement of retained earnings for Sweet Home Catering, Inc., for the month ended March...
-
Journalize the following transactions. Assume the perpetual inventory system. 201X Apr. 5 Sold merchandise for $1,450 cash. The cost of the merchandise was $725. 16 Made refunds to cash customers for...
-
State the statistical notation for each of the following terms: (a) Population size (b) Sample size (c) Population mean (d) Sample mean
-
For the following periodic processes, what is the shortest interval we must examine to see all combinations of deadlines? a. b. c. Process P1 P2 Deadline 2 5 P3 10
-
57. Comprehensive consolidation subsequent to date of acquisition, AAP computation, goodwill, upstream and downstream intercompany inventory profits, downstream intercompany depreciable asset...
-
What is marketing management ? Explain the marketing strategy and plan.
-
Bryant Corporation was incorporated on December 1, 2009, and began operations one week later. Before closing the books for the fiscal year ended November 30, 2010, Bryants controller prepared the...
-
Given a virtual memory system with 32-bit logical addresses and IK Byte pages. The system supports up to 1 GB of physical memory. a) Compute the required number of bits for the page number and for...
-
Monochlorination of pentane, C5H12, leads to formation of how many different products? 2 O1 05
-
In each of the molecules drawn below one chemical bond is colored red. Decide whether this bond is likely to be polar or not. If the bond is likely to be polar, write down the chemical symbol for the...
-
The density of water is approximately 1 g/cm. Which of these objects will float on Water A. Nylon (density = 1.150 g/mL). B. Lithium (density = 0.535 g/mL). OC. Glycerol (density = 1.26 g/mL). D....
-
3. Will the sequence of chemical reactions below accomplish the desired transformations? If the answer is no, identify the steps that are problematic and justify your selections. a) b) OH HO OH 1)...
-
Enter your answer in the provided box. Hydrogen sulfide decomposes according to the following reaction, for which K = 9.30 108 at 700C: 2 HS(g) 2 H(g) + S(g) If 0.49 mol of HS is placed in a 3.0-L...
-
Find the sample size for the problem using given information Population Mean = Mu = 40, sigma = standard deviation=10, sample mean = M= 46, Z value = 3
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
From the electron-pair repulsion model, predict the geometry of the following molecules: a. ClF5 b. SbF5 c. SeF4 d. TeF6
-
A particular microwave oven delivers 800 watts. (A watt is a unit of power, which is the joules of energy delivered, or used, per second.) If the oven uses microwave radiation of wavelength 12.2 cm,...
-
The following equilibrium was studied by analyzing the equilibrium mixture for the amount of H2S produced. A vessel whose volume was 2.50 L was filled with 0.0100 mol of antimony(III) sulfide, Sb2S3,...
-
The following accounts and amounts (balances are normal balances) were taken from the records of Prider Manufacturers Ltd at 30 June 2019. Required (a) Prepare a cost of goods manufactured statement...
-
The following data were taken from the records of Manik Manufacturing Ltd for the year ended 30 June 2019. Required (a) Prepare the cost of goods manufactured schedule for the year ended 30 June...
-
The following demonstration problem illustrates the use of the general journal, the four special journals introduced here, and the general ledger with two subsidiary ledgers. Sidney Carton began...

Study smarter with the SolutionInn App


