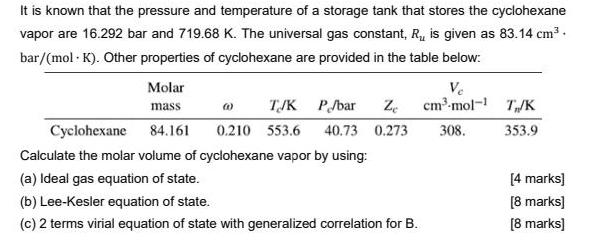
It is known that the pressure and temperature of a storage tank that stores the cyclohexane...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
It is known that the pressure and temperature of a storage tank that stores the cyclohexane vapor are 16.292 bar and 719.68 K. The universal gas constant, R, is given as 83.14 cm3. bar/(mol - K). Other properties of cyclohexane are provided in the table below: Molar Ve cm-mol- T/K mass TJK P/bar Cyclohexane 84.161 0.210 553.6 40.73 0.273 308. 353.9 Calculate the molar volume of cyclohexane vapor by using: (a) Ideal gas equation of state. [4 marks] (b) Lee-Kesler equation of state. [8 marks] (c) 2 terms virial equation of state with generalized correlation for B. [8 marks) It is known that the pressure and temperature of a storage tank that stores the cyclohexane vapor are 16.292 bar and 719.68 K. The universal gas constant, R, is given as 83.14 cm3. bar/(mol - K). Other properties of cyclohexane are provided in the table below: Molar Ve cm-mol- T/K mass TJK P/bar Cyclohexane 84.161 0.210 553.6 40.73 0.273 308. 353.9 Calculate the molar volume of cyclohexane vapor by using: (a) Ideal gas equation of state. [4 marks] (b) Lee-Kesler equation of state. [8 marks] (c) 2 terms virial equation of state with generalized correlation for B. [8 marks)
Expert Answer:

Related Book For 

Introduction to Probability
ISBN: 978-0716771098
1st edition
Authors: Mark Daniel Ward, Ellen Gundlach
Posted Date:
Students also viewed these chemical engineering questions
-
In an auto body shop, it is known that the average number of paint blemishes per car is 3. If 2 cars are randomly chosen for inspection, what is the probability that: (a) The first car has no more...
-
It is known that the pressure and temperature of a storage tank that stores the cyclohexane vapor are 16.292 bar and 719.68 K. The universal gas constant, R, is given as 83.14 cm bar/(mol- K). Other...
-
In a certain federal prison it is known that 2/3 of the inmates are under 25 years of age. It is also known that 3/5 of the inmates are male and that 5/8 of the inmates are female or 25 years of age...
-
Question 4: Partners M, N, and O agreed to share the net income or loss in the ratio of 5:4:3 respectively. Their capital balances were Rs.80, 000, Rs. 70, 000 and Rs. 60, 000. They decided that "O"...
-
Use the information in E11- 13, part ( a) to prepare the required footnote disclosure under IFRS for Kurtis Koal Company, Inc. s property, plant, and equipment for years 1 and 2, including a...
-
A small brewery considers the output of its bottling machine as a continuous income stream with an annual rate of flow at time t given by f (t)=80e-0.1t in thousands of dollars per year. Find the...
-
Light of constant intensity but varying wavelength was used to illuminate the cathode in a photoelectric-effect experiment. The graph of Figure P28.54 shows how the stopping potential depended on the...
-
Indicate whether each of the following is a direct cost (D), an indirect cost (ID), or neither (N) and a variable (V) or a fixed (F) cost. Also indicate whether each adds value (VA) or does not add...
-
2. Continuity and Uniform Continuity (20 Points) Consider the function f(x) = x on the interval [0, 1]. (a) Prove that the function is continuous on [0, 1]. (b) Determine if the function is uniformly...
-
Greyon Inc. operates an active business. Financial statements for the year ended December 31, 2021 report a net income before taxes of $300,000. The following additional information is provided: 1....
-
Using the matrix stiffness method, determine the reactions at the supports of the beam shown in Figure Q2. Take EI to be constant.
-
What do you understand by database. How does it differ from DBMS?
-
What do you understand union compatibility of relations? For which operations such compatibility is required and why?
-
Define a computerised accounting system. Distinguish between a manual and computerised accounting system.
-
From the details given below, find out the credit sales and total sales. Opening debtors Closing debtors Discount allowed Sales returns Irrecoverable amount Bills receivables received Bills...
-
Refer back to the table in Figure 12.7 in the previous chapter. Suppose that aggregate demand increases such that the amount of real output demanded rises by $7 billion at each price level. By what...
-
HBSPWalt Disney Co.: The Entertainment King Examine Disney case. There is a significant amount of information on Disney presented, particularly from 1983 - 2000. what are performance trends in this...
-
(a) Bright Sdn Bhd (BSB) is a tax resident manufacturing company in Johor, which involves in ceramic tiles. Currently, BSBs annual sales turnover has been forecasted to be around RM 300,000 for the...
-
A student makes a trip once per day to the store, and he always buys a snack. The student eats the snack 70% of the time, but the other 30% of the time, his roommate eats it first. a. During a period...
-
Now use the moment generating function from Exercise 43.1 to verify that, if X is Geometric with parameter p, then E(X2) = (2 - p)/p2. In Exercise 1 P(X = x) = px(x) = (1- p)x-lp For x = 1, 2, 3,...,...
-
The time an energy-saving light bulb will last without failing has an Exponential distribution with a median of 3 years. Calculate the probability that a light bulb, selected at random, will last at...
-
A surface with \(N_{0}\) adsorption centers has \(N\left(\leq N_{0}ight)\) gas molecules adsorbed on it. Show that the chemical potential of the adsorbed molecules is given by \[ \mu=k T \ln...
-
Show that for a system in the grand canonical ensemble \[ \{\overline{(N E)}-\bar{N} \bar{E}\}=\left(\frac{\partial U}{\partial N}ight)_{T, V} \overline{(\Delta N)^{2}} \]
-
Study the state of equilibrium between a gaseous phase and an adsorbed phase in a singlecomponent system. Show that the pressure in the gaseous phase is given by the Langmuir equation \[...

Study smarter with the SolutionInn App


