Question: Question 6 a) Apply the work equation to determine the amount of work done by the applied force in each of the three situations
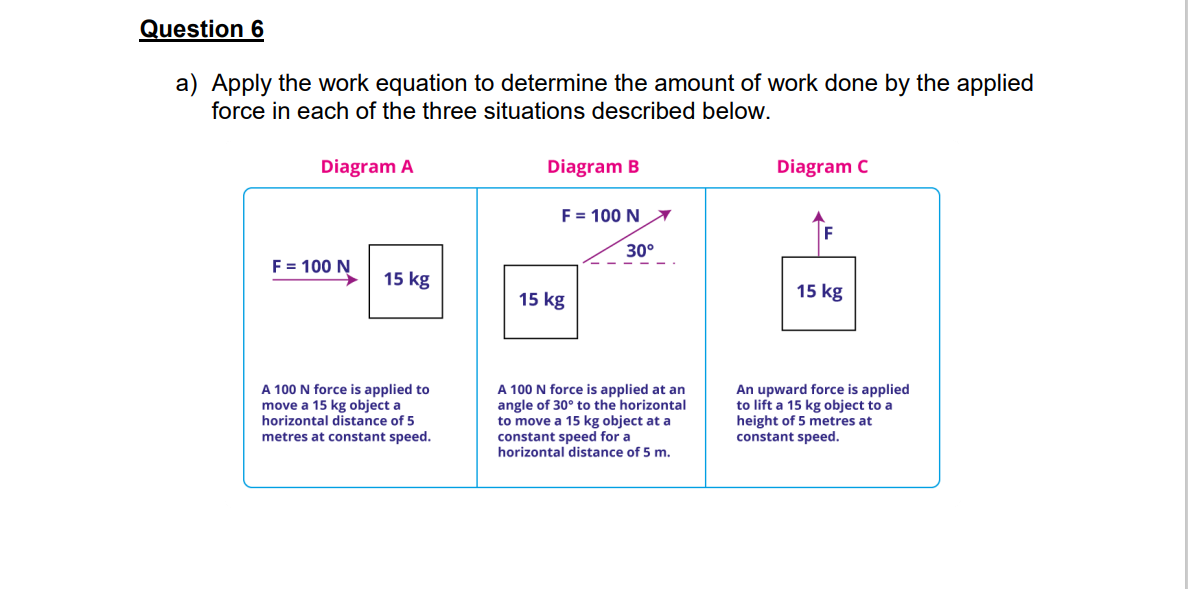



Question 6 a) Apply the work equation to determine the amount of work done by the applied force in each of the three situations described below. Diagram A Diagram B F = 100 N 30 F = 100 N 15 kg 15 kg Diagram C F 15 kg A 100 N force is applied to move a 15 kg object a horizontal distance of 5 metres at constant speed. A 100 N force is applied at an angle of 30 to the horizontal to move a 15 kg object at a constant speed for a horizontal distance of 5 m. An upward force is applied to lift a 15 kg object to a height of 5 metres at constant speed. d) Determine the kinetic energy of a 625kg roller coaster car that is moving with a speed of 18.3m/s. Question 7 (Should you need a copy of the Periodic Table for this question, you will find one on page 8 of this document.) Calculations using Boyle's Law a) 5.00L of a gas is at 1.08atm. What pressure is obtained when the volume is 10.OL? b) 9.48L of a gas was at an unknown pressure. However, at standard pressure, its volume was measured to be 8.00L. What was the unknown pressure? Calculations using Charles' Law c) A gas is collected and found to fill 2.85L at 25.0C. What will be its volume at standard temperature? d) 5.00L of a gas is collected at 100K and then allowed to expand to 20.0L. What must the new temperature be in order to maintain the same pressure? Calculations using Gay-Lussac's Law e) 10.0L of a gas is found to exert 97.0kPa at 25.0C. What would be the required temperature (in Celsius) to change the pressure to standard pressure? f) 5.00L of a gas is collected at 22.0C and 745.0mmHg. When the temperature is changed to standard, what is the new pressure? e) One mole (mol) of any substance consists of 6.022 x 1023 molecules (Avogadro's number). What is the translational kinetic energy of 12.00mol of an ideal gas at 300.OK?
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


