Questions: 1. Calculate the specific heat of your sample. Do this on a piece of paper...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
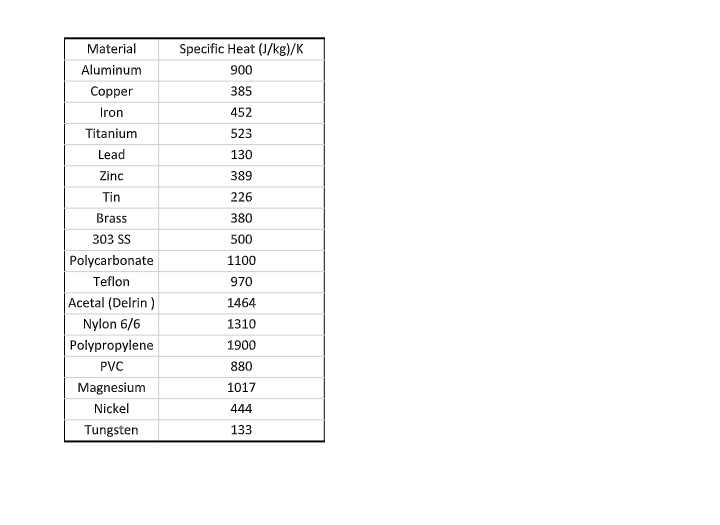
Questions: 1. Calculate the specific heat of your sample. Do this on a piece of paper (or drawpad), take a picture of your work, and include that picture here. 2. What was the specific heat of your sample? 3. Using the table on the next page, determine which material your sample is: object Mo Tia Mw Tin Cw water Co lid Inner Aluminum can Figure: Your Calorimeter setup Experiment: The experimental setup is shown in the above figure: A calorimeter with two cans; the inner aluminum can holds the water and must be accounted for in your calculations, the outer can helps insulate the inner can from the ambient temperature. Cylindrical objects made of an unknown materials will be heated to the temperature of boiling water and then placed in to different inner cans. By measuring the temperature of the boiling water we can closely approximate the temperature of the unknown materials. The sample is quickly removed from the boiler, placed it into the calorimeter, and secured with the lid on the calorimeter. The final temperature of the water is then recorded, which is also the final temperature of the sample and of the aluminum can. Tf Material Aluminum Copper Iron Titanium Lead Zinc Tin Brass 303 SS Polycarbonate Teflon Acetal (Delrin) Nylon 6/6 Polypropylene PVC Magnesium Nickel Tungsten Specific Heat (J/kg)/K 900 385 452 523 130 389 226 380 500 1100 970 1464 1310 1900 880 1017 444 133 Questions: 1. Calculate the specific heat of your sample. Do this on a piece of paper (or drawpad), take a picture of your work, and include that picture here. 2. What was the specific heat of your sample? 3. Using the table on the next page, determine which material your sample is: object Mo Tia Mw Tin Cw water Co lid Inner Aluminum can Figure: Your Calorimeter setup Experiment: The experimental setup is shown in the above figure: A calorimeter with two cans; the inner aluminum can holds the water and must be accounted for in your calculations, the outer can helps insulate the inner can from the ambient temperature. Cylindrical objects made of an unknown materials will be heated to the temperature of boiling water and then placed in to different inner cans. By measuring the temperature of the boiling water we can closely approximate the temperature of the unknown materials. The sample is quickly removed from the boiler, placed it into the calorimeter, and secured with the lid on the calorimeter. The final temperature of the water is then recorded, which is also the final temperature of the sample and of the aluminum can. Tf Material Aluminum Copper Iron Titanium Lead Zinc Tin Brass 303 SS Polycarbonate Teflon Acetal (Delrin) Nylon 6/6 Polypropylene PVC Magnesium Nickel Tungsten Specific Heat (J/kg)/K 900 385 452 523 130 389 226 380 500 1100 970 1464 1310 1900 880 1017 444 133
Expert Answer:
Answer rating: 100% (QA)
Specific Heat Formula When heat energy is added to a substance the temperature will change by a certain amount The relationship between heat energy an... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A technician measures the specific heat of an unidentified liquid by immersing an electrical resistor in it. Electrical energy is converted to heat transferred to the liquid for 120 s at a constant...
-
What is the specific heat of a metal substance if 135 kJ of heat is needed to raise 5.1 kg of the metal from 18.0oC to 31.5oC?
-
What is the specific heat of a metal substance if 135 kJ of heat is needed to raise 4.1 kg of the metal from 18.0C to 37.2C?
-
Josh Kuchin is studying for the next accounting mid-term examination. What should Josh know about? (a) Departing from the cost basis of accounting for inventories (b) The meaning of "market" in the...
-
Sleep apnea is a common sleep disorder characterized by collapses of the upper airway during sleep. Chance (Winter 2009) investigated the role of sleep apnea in how people transition from one sleep...
-
John Parsons (123-45-6781) and George Smith (123-45-6782) are 70% and 30% owners, respectively, of Premium, Inc. (11-1111111), a candy company located at 1005 16th Street, Cut and Shoot, TX 77303....
-
Each message in a digital communication system is classified as to whether it is received within the time specified by the system design. If three messages are classified, use a tree diagram to...
-
Ramakrishnan Inc. reported 2018 net income of $15 million and depreciation of $2,650,000. The top part of Ramakrishnan, Inc.'s 2018 and 2017 balance sheets is listed below (in millions of dollars)....
-
Effects of Adjusting Entries on the Accounting Equation Four adjusting entries are shown below. a. Interest Expense 1,595 Interest Payable 1,595 b. Interest Receivable 1,050 Interest Income 1,050 c....
-
A partially completed worksheet for At Home Pet Grooming Service, a firm that grooms pets at the owner's home, follows. INSTRUCTIONS 1. Record balances as of December 31 in the ledger accounts. 2....
-
Based on the latest financial statements and other financial data of the SCS Corporation presented below, calculate the following: a) Free cash flows (30 points) b) Enterprise Value (10 points) c)...
-
Find the amount of the balance forward ( in $ ) that would result following these transactions. starting balance: $ 2 , 4 3 6 . 8 0 May 2 ; check # 7 9 1 ; to Dreamscape Landscaping; amount of $ 3 3...
-
K James purchases a $20,000 6-month certificate of deposit. Is this counted in M1 only, M2 only, M1 and M2, or neither?
-
Are families disintegrating? Breaking down? Explain your answer and reasoning in a detailed paragraph. Thanks.
-
What are the different family forms and how they have changed over time? What information on the different parenting practices and how they are influenced by the society we live in? the different...
-
Prepare an income statement through gross profit for June 2 0 2 5 assuming sales revenue is $ 1 1 6 , 9 0 0 . MARIN CORPORATION Income Statement ( Partial ) Marin Corporation has the following cost...
-
Consider the following variant for the Tower of Hanoi problem. Suppose there are four needles A, B, C, D. Initially all n disks are at A and they have to be moved such that the top n/2 end up at C...
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
An air bubble at the bottom of a lake 41.0 m deep has a volume of 1.00 cm3. If the temperature at the bottom is 5.5C and at the top 18.5C, what is the radius of the bubble just before it reaches the...
-
1. A square loop moves to the right from an area where B = 0 completely through a region containing a uniform magnetic field directed into the page (Fig. 21-52), and then out to B = 0 after point L....
-
Determine the activities of (a) 1.0 g of 13153I (T12 = 8.02 days) (b) 1.0 g of 23892U (T12 = 4.47 109 yr)
-
(a) The longitudinal data set "v4c" is in the vertical format; transform it into the horizontal format. (b) Transform the data set you obtained in part (a) back into the vertical format.
-
Perform some exploratory analysis on the DTS study described above. (a) Compute the mean and standard deviation of the HamD scores for the two treatment groups at each time point. (b) Treat repeated...
-
In this question we develop a regression model to assess the treatment effect for stigma in the DTS study, controlling for demographics and baseline measurements. We will use the cumulative logit...

Study smarter with the SolutionInn App


