questions. If the solution of u + +2 + + =0 is of the form u(x,y)=e***,...
Fantastic news! We've Found the answer you've been seeking!
Question:
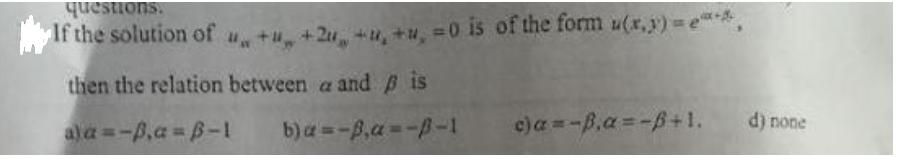
Transcribed Image Text:
questions. If the solution of u + +2 + + =0 is of the form u(x,y)=e***, then the relation between a and is a) a=-p.a=B-1 b) a=-ß,a=-B-1 c)a=-B,a=-ß+1. d) none questions. If the solution of u + +2 + + =0 is of the form u(x,y)=e***, then the relation between a and is a) a=-p.a=B-1 b) a=-ß,a=-B-1 c)a=-B,a=-ß+1. d) none questions. If the solution of u + +2 + + =0 is of the form u(x,y)=e***, then the relation between a and is a) a=-p.a=B-1 b) a=-ß,a=-B-1 c)a=-B,a=-ß+1. d) none
Expert Answer:
Answer rating: 100% (QA)
The image youve shared presents a question from a mathematics problem set related to linear homogene... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these accounting questions
-
In a PowerPoint presentation of 4-6 slides: Identify and briefly describe your personal conflict-management style. Explain how to work with the selected conflict-management style mentioned above....
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A student comes to the professor's office to say that her group did not get the group assignment finished. She says that one member of the group of four is not carrying his fair share of the load and...
-
If the interest rate on debt is lower than ROA, then a firm will Valitse yksi: a. increase ROE b. not change ROE c. none of these is correct d. decrease ROE e. change the ROE in an indeterminable...
-
Refer to the Carleton University balanced scorecard in Exhibit 17.8. The full version of the scorecard is available on the Carleton University website: www5.carleton.ca/fi...
-
Companies are aware that analysts focus on profitability in evaluating financial performance. Managers have historically utilized a number of methods to improve reported profitability that are...
-
A confidence interval for a mean response and a prediction interval for an individual response are to be constructed from the same data. True or false: The number of degrees of freedom for the...
-
Tip Top Company sells umbrellas suited for small and large picnic tables. Based on the advice of its accountant, Tip Top is considering whether to adopt an activity-based costing system. To evaluate...
-
What are the challenges associated with managing cultural diversity within multicultural organizations, and how can organizations foster an inclusive culture that celebrates diversity while...
-
Solve Prob. 411 using singularity functions. Use statics to determine the reactions. Prob. 411, A simply supported beam loaded by two forces is shown in the figure. Select a pair of structural steel...
-
A is a shareholder in X Co., an S corporation. In 20x3, A's share of X Co.'s net operating loss is $40,000. On 12/3/x3, A has a basis in his X Co. stock of $10,000. A also has a loan due from X Co....
-
You have opened an account with a local broker & you placed an order for 4,000.0 shares of KLM corp. @ $8.0 a share. The initial margin is 50.0%; the broker's rate is 6.0% and the maintenance margin...
-
How I Communicate ? Think about your day yesterday. List 10 people with whom you communicated and what type of communication was established. The communication may be verbal, non-verbal, written,...
-
A chemical reaction can absorb or release energy, forming endothermic or exothermic reaction. Explain why a chemical reaction becomes exothermic or endothermic in nature using suitable examples.
-
Why are chemical reactions and chemical changes significant for the world? How wouldn't humans exist if it wasn't for chemical reactions and chemical changes?
-
5 Shadee Corporation expects to sell 520 sun shades in May and 440 in June. Each shade sells for $140. Shadee's beginning and ending finished goods inventories for May are 85 and 50 shades,...
-
Problem 7. Recall that the standard basis of R is = [e e2] where - 0 -8 Let where and let 1 where U = [ ], -13]. U = U1 U12 V = [v v], 3. 0 Then U and V are ordered bases of R2 (you don't need to...
-
(a) How far away can a human eye distinguish two ear headlights 2.0 m apart? Consider only diffraction effects and assume an eye pupil diameter of 5.0 mm and a wavelength of 550 nm. (b) What is the...
-
Kent Pham, CPA, is a 45-year-old single taxpayer living at 169 Trendie Street, La Jolla, CA 92037. His Social Security number is 865-68-9635. In 2012, Kent's W-2 as the controller of a local...
-
Clarita is a single taxpayer with two dependent children, ages 10 and 12. Clarita pays $3,000 in qualified child care expenses during the year. If her adjusted gross income (all from wages) for the...
-
Diane is a single taxpayer who qualifies for the earned income credit. Diane has two qualifying children who are 3 and 5 years old. During 2012, Diane's wages are $17,100 and she receives dividend...
-
Ammonia synthesis reaction is represented by \[ \mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) ightarrow 2 \mathrm{NH}_{3}(\mathrm{~g}) \] Initially, \(1 \mathrm{~mol}\) of nitrogen and...
-
A mixture of \(1 \mathrm{~mol}\) of sulphur dioxide gas, \(0.5 \mathrm{~mol}\) of oxygen gas and \(2 \mathrm{~mol}\) of argon gas are fed into a reactor at \(30 \mathrm{bar}\) and \(900 \mathrm{~K}\)...
-
The dissociation of phosphorus pentachloride takes place at \(250^{\circ} \mathrm{C}\) as \[ \mathrm{PCl}_{5} ightleftharpoons \mathrm{PCl}_{3}+\mathrm{Cl}_{2} \quad K_{P}=1.8 \] Calculate the...

Study smarter with the SolutionInn App


