Ray Company provided the following excerpts from its Production Department's flexible budget performance report. (Round rate...
Fantastic news! We've Found the answer you've been seeking!
Question:
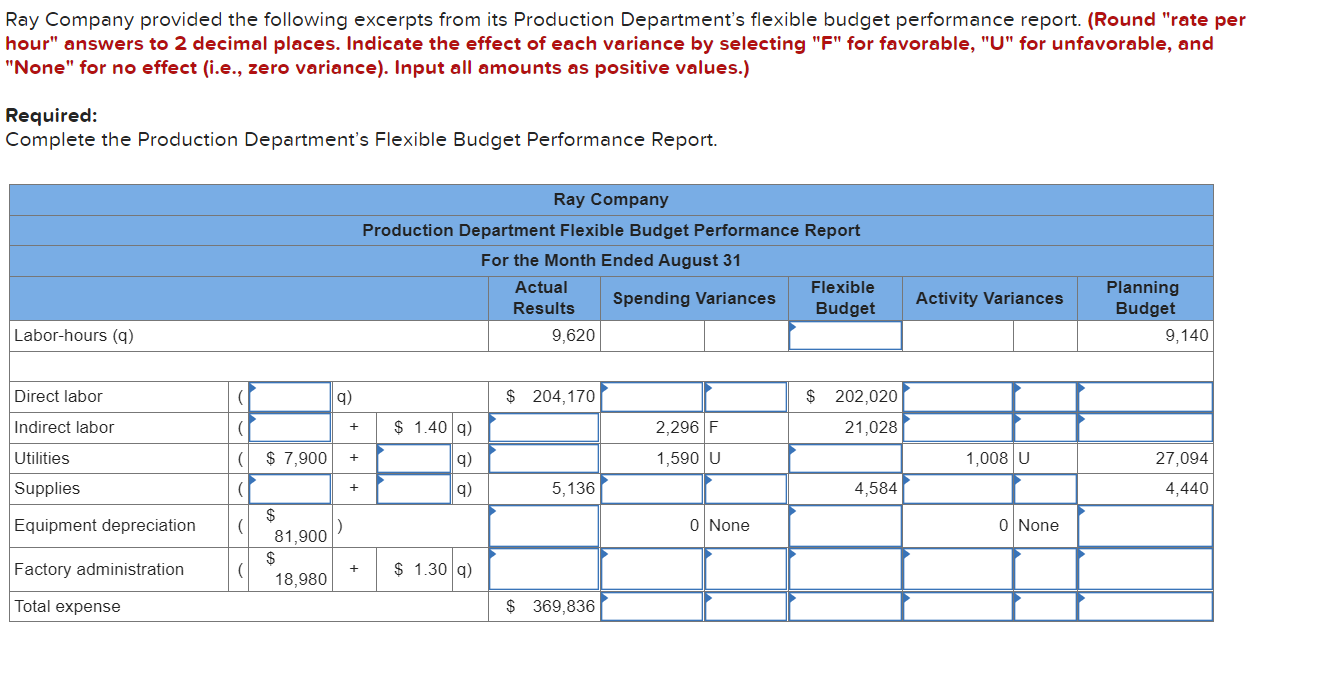
Transcribed Image Text:
Ray Company provided the following excerpts from its Production Department's flexible budget performance report. (Round "rate per hour" answers to 2 decimal places. Indicate the effect of each variance by selecting "F" for favorable, "U" for unfavorable, and "None" for no effect (i.e., zero variance). Input all amounts as positive values.) Required: Complete the Production Department's Flexible Budget Performance Report. Ray Company Production Department Flexible Budget Performance Report For the Month Ended August 31 Actual Results Spending Variances Flexible Budget Activity Variances Planning Budget Labor-hours (q) 9,620 Direct labor ( q) $ 204,170 $ 202,020 Indirect labor ( 21,028 2,296 F 1,590 U Utilities ( $ 7,900 1,008 U Supplies ( 5,136 4,584 $ 81,900 Equipment depreciation ( ( Factory administration $ 18,980 Total expense $ 369,836 ) $ 1.40 q) q) q) + $ 1.30 q) + + + 0 None 0 None 9,140 27,094 4,440 Ray Company provided the following excerpts from its Production Department's flexible budget performance report. (Round "rate per hour" answers to 2 decimal places. Indicate the effect of each variance by selecting "F" for favorable, "U" for unfavorable, and "None" for no effect (i.e., zero variance). Input all amounts as positive values.) Required: Complete the Production Department's Flexible Budget Performance Report. Ray Company Production Department Flexible Budget Performance Report For the Month Ended August 31 Actual Results Spending Variances Flexible Budget Activity Variances Planning Budget Labor-hours (q) 9,620 Direct labor ( q) $ 204,170 $ 202,020 Indirect labor ( 21,028 2,296 F 1,590 U Utilities ( $ 7,900 1,008 U Supplies ( 5,136 4,584 $ 81,900 Equipment depreciation ( ( Factory administration $ 18,980 Total expense $ 369,836 ) $ 1.40 q) q) q) + $ 1.30 q) + + + 0 None 0 None 9,140 27,094 4,440
Expert Answer:
Answer rating: 100% (QA)
Actual Spending variance Flexible Activity variance Planning results budget budget labor hours ... View the full answer

Related Book For 

Managerial Accounting
ISBN: 978-1259307416
16th edition
Authors: Ray Garrison, Eric Noreen, Peter Brewer
Posted Date:
Students also viewed these accounting questions
-
Ray Company provided the following excerpts from its Production Department's flexible budget performance report: Required: Complete the Production Department's Flexible Budget Performance Report by...
-
Ray Company provided the following excerpts from its Production Department's flexible budget performance report. Required: Complete the Production Department's Flexible Budget Performance Report....
-
For 2012, Ontario Manufacturing Company provided the following accounting information: a. Which of the above are sources of funds? b. Which of the above are uses of funds? c. What is the overall...
-
The light bulbs produced by Edison Lighting Corporation last an average of 300 h. The life of the light bulbs is believed to follow a normal distribution with a standard deviation of 10. A customer...
-
No More Books Corporation has an agreement with Floyd Bank, whereby the bank handles $3.2 million in collections a day and requires a $350,000 compensating balance. No More Books is contemplating...
-
The following table indicates the amounts of Co and S before reaction. Complete the table after reactions (a) and (b) according to the balanced equation: 2 Co(s) + 3 S(s) C0S3(s)
-
What does a report on personal financial statements typicallv address?
-
Norrington Trading Co. provides the following income statement for 2011: Sales . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
you purchased a new washer and dryer for $123.999including salestax and delivery). Financing was used for the fullpurchase price and the payments are $144.84 per month for 11months. what is the cost...
-
1. What is it about Rachel's leadership that clearly suggests that she is engaged in transformational leadership? 2. In what ways has the growth of ICCR had an impact on Rachel's leadership? 3. Given...
-
Determine the net distributable estate. M, non-resident, married to N, died in 2018 leaving the following: a. Real properties in Pampanga- conjugal b. Real property abroad- exclusive (M) c. Real...
-
With whom do you have the most troublesome conflicts? What are your conflicts with these parties about? What have you noticed about the different ways in which men and women engage conflict? Why is...
-
Why is it so distressing to find out someone has lied to you? In what professions do you think people lie the most? The least? How do you feel when you lie to someone? In what situations are you the...
-
Explain the role of system call interfaces in operating systems. How do they ensure a secure boundary between user space and kernel space ?
-
Question: Equipment was purchased for $36,000 by issuing a 5-year note payable to the seller for $20,000, the balance paid in cash. The note pays 5% interest annually on January 1 st. For expense...
-
There are cultures that believe eye contact is disrespectful. Name at least one (1) of those cultures, its location in this world, and the reasons why eye contact is a sign of disrespect. Why do you...
-
Use the following information to calculate the non-dilutive exchange ratio [EPS (Gillette)= $1.60 and EPS (P&G) = $1.88]. Is P&G's offer dilutive or accretive? Show your calculation.
-
How do individual companies respond to economic forces throughout the globe? One way to explore this is to see how well rates of return for stock of individual companies can be explained by stock...
-
Explain the four-step process used to compute a predetermined overhead rate.
-
Distinguish between discretionary fixed costs and committed fixed costs.
-
What are unit-level activities? Batch-level activities? Product-level activities? Facility-level activities?
-
Estimate the depression in the freezing point if \(58.5 \mathrm{~g}\) of \(\mathrm{NaCl}\) is added to \(1 \mathrm{~L}\) of water at atmospheric pressure.
-
Calculate the molal boiling point elevation constant of benzene if its heat of vaporization at \(80.1^{\circ} \mathrm{C}\) is \(30.67 \mathrm{~kJ} / \mathrm{mol}\).
-
A hydrocarbon \(\mathrm{H}_{2}\left(\mathrm{CH}_{2} ight)_{n}\) is dissolved in a solvent \(S\) which freezes at \(9.0^{\circ} \mathrm{C}\). A solution which contains \(0.90 \mathrm{~g}\) of...

Study smarter with the SolutionInn App


