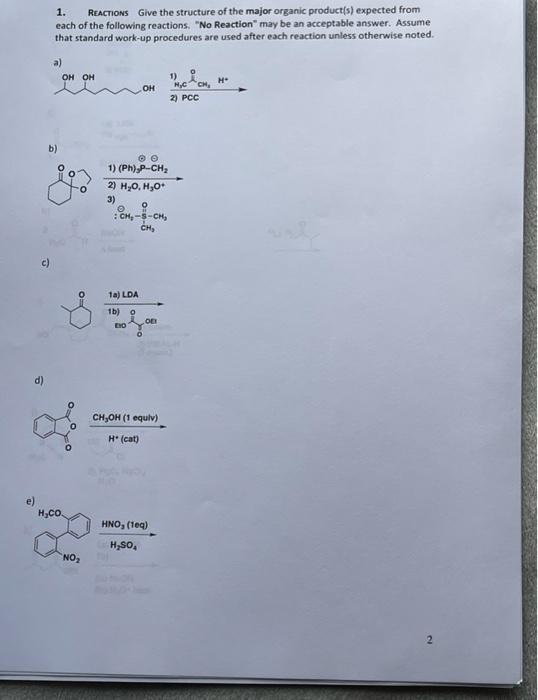
REACTIONS Give the structure of the major organic product(s) expected from each of the following reactions,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
REACTIONS Give the structure of the major organic product(s) expected from each of the following reactions, "No Reaction" may be an acceptable answer. Assume that standard work-up procedures are used after each reaction unless otherwise noted. 1. a) он он 1) H* он 2) PCC b) 1) (Ph),P-CH; 2) H,0, H,0+ 3) CH, c) 1a) LDA 1b) o (P. CH,OH (1 equiv) H (cat) e) H,CO. HNO, (teg) H,sO, NO REACTIONS Give the structure of the major organic product(s) expected from each of the following reactions, "No Reaction" may be an acceptable answer. Assume that standard work-up procedures are used after each reaction unless otherwise noted. 1. a) он он 1) H* он 2) PCC b) 1) (Ph),P-CH; 2) H,0, H,0+ 3) CH, c) 1a) LDA 1b) o (P. CH,OH (1 equiv) H (cat) e) H,CO. HNO, (teg) H,sO, NO
Expert Answer:
Related Book For 

Organic Chemistry A Short Course
ISBN: 978-1111425562
13th edition
Authors: Harold Hart, Christopher M. Hadad, Leslie E. Craine, David J. Hart
Posted Date:
Students also viewed these chemistry questions
-
Give the structure of the major alkene formed when the hydroxide of each of the following quaternary ammonium ions is heated.
-
Write the structure of the major organic product obtained by hydroboration-oxidation of each of the following alkenes: (a) 2-Methylpropene (b) cis-2-Butene (c) (d) Cyclopentene (e) 3-Ethyl-2-pentene...
-
Write the structure of the major organic product formed in the reaction of 1-pentene with each of the following: (a) Hydrogen chloride (b) Hydrogen bromide (c) Hydrogen bromide in the presence of...
-
Pacific Company sells electronic test equipment that it acquires from a foreign source. During the year 2014, the inventory records reflected the following: Inventory is valued at cost using the LIFO...
-
Access the IRS Internet site atwww.irs.gov and answer the following questions: a. How does one file a tax return electronically? b. How can the taxpayer transmit funds electronically? c. What are the...
-
Using the molecular orbital model to describe the bonding in CO, CO+, and CO2+, predict the bond orders and the relative bond lengths for these three species. How many unpaired electrons are present...
-
Bernardino Santos-Rodiguez was piloting a boat owned by his friend, Raul Viera-Torres, in waters near Guayama, Puerto Rico. The boat was equipped with a hydraulic steering system manufactured by...
-
Sales of industrial vacuum cleaners at R. Lowenthal Supply Co. over the past 13 months are as follows: (a) Using a moving average with three periods, determine the demand for vacuum cleaners for next...
-
(a) 1. Assume a Heckscher-Ohlin world. The two countries are China and the U.S. The two goods are wheat and cloth and the two factors are labor and capital. Suppose each ton of wheat requires 4 units...
-
Design a steam power cycle that can achieve a cycle thermal efficiency of at least 40 percent under the conditions that all turbines have isentropic efficiencies of 85 percent and all pumps have...
-
Felix has already sold 2 tubs, Brody hasn't sold any yet. If Felix starts selling 3 tubs per day and Brody begins selling 5 tubs per day, they will eventually sell the same amount of cookie dough....
-
Identify two target customers. For ONE target customer describe 2-3 different persona based on data and research. Identify common pain points these personas might experience based on research and...
-
Definitions of the standard cost card and the two types of standards Comparison of master budgets versus flexible budgets Definitions of volume variance and spending variance Responsible persons for...
-
Example of a product which did aOptional-product pricing.Discuss Example of a product which did aCaptive-Product Pricing.Discuss
-
Following this NUT 10V quiz, you decide to make yourself a big bowl of bacon mac and cheese. TOTAL fat for this meal is 72 grams. Based on the 2020 Dietary Guidelines, what percent of the Daily Value...
-
Go to the Internet site of the state courts in your state. (See Appendix D.) Find the court rules of any civil court in your state that hears divorce cases. Quote from any court rule that uses the...
-
Vulcan Flyovers offers scenic overflights of Mount Saint Helens, the volcano in Washington State that explosively erupted in 1982. Data concerning the company's operations in July appear below:...
-
Reread the discussion leading to the result given in (7). Does the matrix sI - A always have an inverse? Discuss.
-
Divide the following compounds into groups that might be expected to exhibit similar chemical behavior: a. C4H10 b. CH3OCH3 c. C3H7OH d. C8H18 e. HOCH2CH2CH2OH f. CH3NH2 g. CH3CH2CH3 h. CH3OH i....
-
Identify the hydrogens that are readily exchanged for deuterium in CH2CH3 b. (CHs) CCCH a.
-
The RNA base uracil is a pyrimidine derivative, usually depicted in its keto form (page 396). The enol tautomer readily undergoes electrophilic substitution, unlike most pyrimidines. a. Draw the...
-
Kids Sports Consulting Pty Ltd is a company set up by sports and recreation management students to gain experience in running their own business. It had the following contribution margin income...
-
V. Zarb, the marketing manager for Maltese Treasures Ltd, is preparing a sales budget for the year ended 30 June 2020. In reviewing the actual sales data for the previous year, the sales and...
-
The following expenses budget has been prepared for Abacus Services for the year ending 30 June 2020. Professional salaries, secretarial wages and training are paid in the quarter in which they are...

Study smarter with the SolutionInn App



