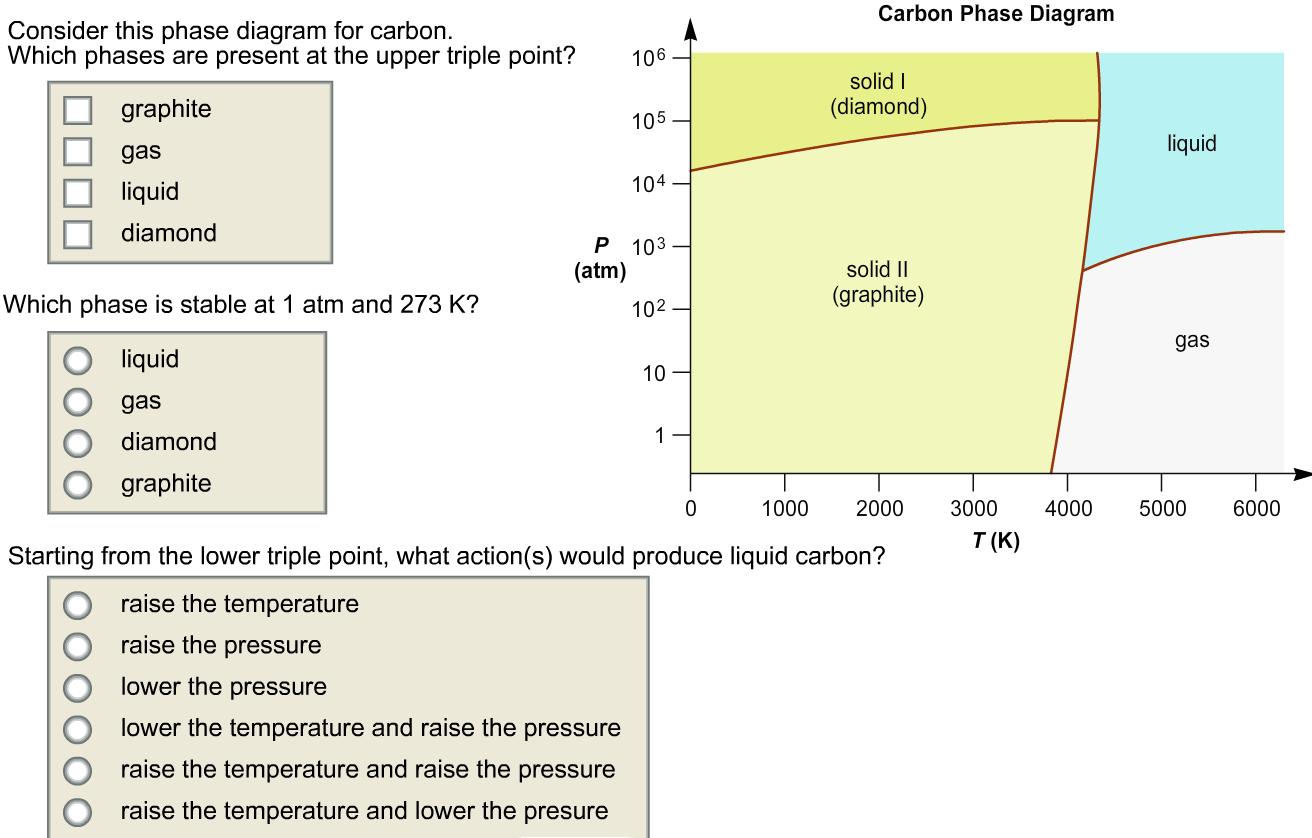
Consider this phase diagram for carbon. Which phases are present at the upper triple point? graphite...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider this phase diagram for carbon. Which phases are present at the upper triple point? graphite gas liquid diamond Which phase is stable at 1 atm and 273 K? liquid gas diamond graphite (atm) 106 105. P 103 lower the pressure lower the temperature and raise the pressure raise the temperature and raise the pressure raise the temperature and lower the presure 104 10² 10 1 Carbon Phase Diagram solid I (diamond) solid II (graphite) 0 Starting from the lower triple point, what action(s) would produce liquid carbon? raise the temperature raise the pressure 1000 2000 liquid gas 3000 4000 5000 T(K) 6000 Consider this phase diagram for carbon. Which phases are present at the upper triple point? graphite gas liquid diamond Which phase is stable at 1 atm and 273 K? liquid gas diamond graphite (atm) 106 105. P 103 lower the pressure lower the temperature and raise the pressure raise the temperature and raise the pressure raise the temperature and lower the presure 104 10² 10 1 Carbon Phase Diagram solid I (diamond) solid II (graphite) 0 Starting from the lower triple point, what action(s) would produce liquid carbon? raise the temperature raise the pressure 1000 2000 liquid gas 3000 4000 5000 T(K) 6000
Expert Answer:
Answer rating: 100% (QA)
TRIPLE POINT THREE 1 2 Ans 3 PHASES PHASES OF UPPER AT AT TRIPLE POINT ANS PRESENT UPPER ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these chemistry questions
-
Saturated water vapor at 400F is heated at constant pressure until its volume has doubled. Determine the final temperature using the ideal gas equation of state, the compressibility charts, and the...
-
Ethylene is heated at constant pressure from 5 MPa and 20oC to 200oC. Using the compressibility chart, determine the change in the ethylene's specific volume as a result of this heating.
-
Suppose 1.00 mol of oxygen is heated at constant pressure of 1.00 atm from 10.0C to 25.0C. (a) How much heat is absorbed by the gas? (b) Using the ideal gas law, calculate the change of volume of the...
-
At Blossom Company, events and transactions during 2020 included the following. The tax rate for all items is 20%. (1) Depreciation for 2018 was found to be understated by $148000. (2) A strike by...
-
A tire manufacturer wants to determine the inner diameter of a certain grade of tire. Ideally, the diameter would be 570 mm. The data are as follows: 572,572,573,568,569,575,565,570. (a) Find the...
-
Nitrogen at 120 kPa and 30C is compressed to 600 kPa in an adiabatic compressor. Calculate the minimum work needed for this process in kJ/kg. 600 kPa Nitrogen compressor 120 kPa 30C
-
What format to use for stating a cause of action?
-
Beck Company set the following standard unit costs for its single product. Direct materials (26 Ibs. @ $4 per Ib.) . . . . . . . . . . . . . . . . $104.00 Direct labor (8 hrs. @ $8 per hr.) . . . . ....
-
Let u= (3, 5, 1) and v = (2, -2, 3). Find 8u + 7v = i 8u + 7v. i ).
-
Orion Controls is a leading manufacturer of industrial valve systems, and Nathan Armstrong, head of Marketing, had been contacted by Andre Gide, EVP of Avion Chemical to place an order for 50 of...
-
Sandhill Co. started 3,600 units during the period. Its beginning inventory is 600 units one-fourth complete as to conversion costs and 100% complete as to materials costs. Its ending inventory is...
-
What are the differences among open outcry auctions, dealer markets, and automated trading platforms with automated matching engines?
-
Identify two kinds of lawsuits brought by current or former employees that may result from a negative reference.
-
Explain the difference between marginal tax rates and average tax rates.
-
Describe some of the motives and mistakes made by the Fed, home buyers, mortgage brokers, real estate appraisers, mortgage originators, mortgage securitizers, financial engineers, and investors.
-
Indicate whether each of the following statements is true or false by writing T or F in the answer c olumn. Employees who are victims of illegal discrimination may recover punitive da mages.
-
Describe the escalation of commitment bias, and give an example of it an action. Identify and describe four tactics that managers can use to reduce this bias?
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
The pressure gage on a 2.5-m3 oxygen tank reads 500 kPa. Determine the amount of oxygen in the tank if the temperature is 28oC and the atmospheric pressure is 97 kPa. 2 500 kPa 02 V 25 m T 28C FIGURE...
-
A gas mixture consists of 6 kmol of H2 and 2 kmol of N2. Determine the mass of each gas and the apparent gas constant of the mixture.
-
Methane (CH4) is burned with stoichiometric amount of air during a combustion process. Assuming complete combustion, determine the air-fuel and fuel-air ratios.
-
Determine the principal stresses acting at point \(A\) of the supporting frame. Show the results on a properly oriented element located at this point. B 150 mm -800 mm- 300 mm 12 mm B 130 mm- -15 mm...
-
Determine the principal stresses acting at point \(B\), which is located just on the web, below the horizontal segment on the cross section. Show the results on a properly oriented element located at...
-
Determine the principal stresses in the box beam at point \(B\). Show the results on an element located at this point. 10 kip 4 kip A B* -2 ft- -1.5 ft -2 ft- ft- 0.5 ft 4 in. HA in. 4 in. B 6 in. 3...

Study smarter with the SolutionInn App


