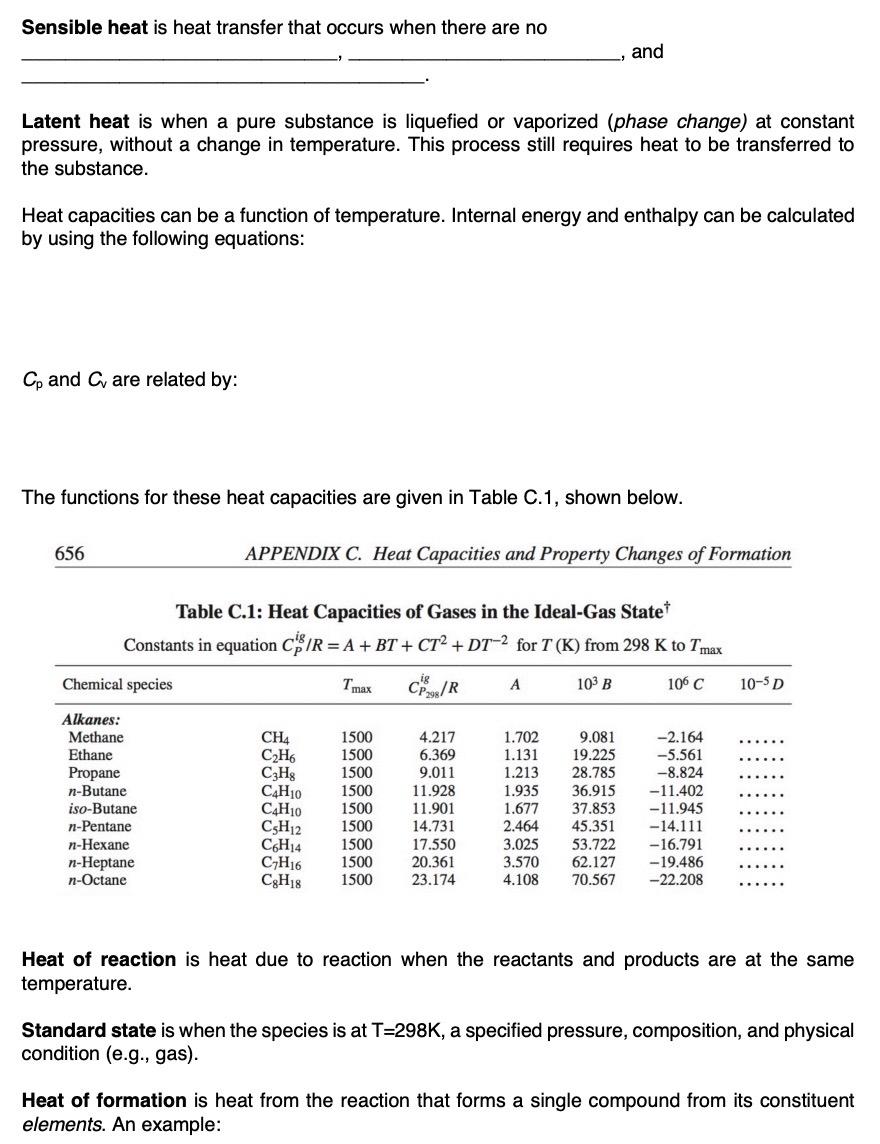
Sensible heat is heat transfer that occurs when there are no and Latent heat is when...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Sensible heat is heat transfer that occurs when there are no and Latent heat is when a pure substance is liquefied or vaporized (phase change) at constant pressure, without a change in temperature. This process still requires heat to be transferred to the substance. Heat capacities can be a function of temperature. Internal energy and enthalpy can be calculated by using the following equations: Cp and C are related by: The functions for these heat capacities are given in Table C.1, shown below. 656 APPENDIX C. Heat Capacities and Property Changes of Formation Table C.1: Heat Capacities of Gases in the Ideal-Gas State* Constants in equation C/R = A + BT + CT + DT-2 for T (K) from 298 K to Tmax Chemical species Tmax ig CP298/R A 103 B 106 C 10-5 D Alkanes: Methane CH4 1500 4.217 1.702 9.081 -2.164 Ethane C2H6 1500 6.369 1.131 19.225 -5.561 Propane C3H8 1500 9.011 1.213 28.785 -8.824 n-Butane C4H10 1500 11.928 1.935 36.915 -11.402 iso-Butane C4H10 1500 11.901 1.677 37.853 -11.945 n-Pentane C5H12 1500 14.731 2.464 45.351 -14.111 n-Hexane C6H14 1500 17.550 3.025 53.722 -16.791 n-Heptane C7H16 1500 20.361 3.570 62.127 -19.486 n-Octane C8H18 1500 23.174 4.108 70.567 -22.208 Heat of reaction is heat due to reaction when the reactants and products are at the same temperature. Standard state is when the species is at T-298K, a specified pressure, composition, and physical condition (e.g.. gas). Heat of formation is heat from the reaction that forms a single compound from its constituent elements. An example: Sensible heat is heat transfer that occurs when there are no and Latent heat is when a pure substance is liquefied or vaporized (phase change) at constant pressure, without a change in temperature. This process still requires heat to be transferred to the substance. Heat capacities can be a function of temperature. Internal energy and enthalpy can be calculated by using the following equations: Cp and C are related by: The functions for these heat capacities are given in Table C.1, shown below. 656 APPENDIX C. Heat Capacities and Property Changes of Formation Table C.1: Heat Capacities of Gases in the Ideal-Gas State* Constants in equation C/R = A + BT + CT + DT-2 for T (K) from 298 K to Tmax Chemical species Tmax ig CP298/R A 103 B 106 C 10-5 D Alkanes: Methane CH4 1500 4.217 1.702 9.081 -2.164 Ethane C2H6 1500 6.369 1.131 19.225 -5.561 Propane C3H8 1500 9.011 1.213 28.785 -8.824 n-Butane C4H10 1500 11.928 1.935 36.915 -11.402 iso-Butane C4H10 1500 11.901 1.677 37.853 -11.945 n-Pentane C5H12 1500 14.731 2.464 45.351 -14.111 n-Hexane C6H14 1500 17.550 3.025 53.722 -16.791 n-Heptane C7H16 1500 20.361 3.570 62.127 -19.486 n-Octane C8H18 1500 23.174 4.108 70.567 -22.208 Heat of reaction is heat due to reaction when the reactants and products are at the same temperature. Standard state is when the species is at T-298K, a specified pressure, composition, and physical condition (e.g.. gas). Heat of formation is heat from the reaction that forms a single compound from its constituent elements. An example:
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Portray in words what transforms you would have to make to your execution to some degree (a) to accomplish this and remark on the benefits and detriments of this thought.You are approached to compose...
-
Markku Antero Oy produces perfume. To make this perfume, Markku Antero uses three different types of fluid. Tartarus, Erebus and Uranus are used in standard proportions of -14-0, -3- 10 and -3- 10,...
-
Design a T network necessary to realize the following z parameters at = 106 rad/s 4t/3 2 5-j
-
How can training stakeholders energize and sustain active learning processes?
-
At the equator, the direction of Earth's magnetic field is horizontal and to the north, and the magnitude is \(3.5 \times 10^{-5} \mathrm{~T}\). (a) What is the magnetic flux through a circular loop...
-
Suppose the exchange rate between U.S. dollars and the Swiss franc was SFr1.6 = $1, and the exchange rate between the dollar and the British pound was 1 = $1.50. What was the exchange rate between...
-
12. "New competition is not between what companies produce in their factories, but between what they add to their factory output in the form of packaging, services, advertising, customer advice,...
-
A product and service are bundled together and sold to customers for $450. The fair values of the product and service are $350 and $150 respectively. Under the residual method, how much would be...
-
A portfolio consists two securities as listed in the following table, assuming that the correlation coefficient is 0.5. Use the given information to answer the following two questions. Securities...
-
a) Construct a graph of Weight versus Spring Displacement with Spring Displacement on the x-axis. Draw the line that best fits your data points. The slope of the graph is the spring constant for the...
-
Juanita Torres is 35-years old and lately has been working with her financial planner. She is attempting develop a long-term savings and investment program. She has been thinking about all the major...
-
Evaluate the following definite integral using the method of Example 10.2 (page 67): 90 0 x +1 dx for a real with a > 0. Hint: Since the integrand is an even function of x you may use * cos(ax) dx...
-
There are two horizontal rows of four students each facing each other. The rows are called Row-1 and Row-2. Eight students are named from A to H. The following conditions apply: 1. A stands opposite...
-
ABC Company has prepared the following purchases budget: All purchases are paid for as follows: 10% in the month of purchase, 50% in the following month, and 40% two months after purchase. Calculate...
-
APC16550D UART has a clock running at18.432 MHz and its baud rate is set to 2000.Determine the HEX contents of its DLM and DLL registers. Please can you explain step by step and in detail how you get...
-
Which of the following function(s) is/are carried out by piRITS or piRISC? a. Inhibits transcription of TEs b. Causes the degradation of TE RNA c. Causes chromosome breakage d. Both a and b are...
-
Scientists propose that the first macromolecules in protobionts were a. DNA molecules. b. RNA molecules. c. proteins. d. all of the above.
-
In the CRISPR-Cas system, what does tracrRNA bind to? a. crRNA and Cas1 protein b. crRNA and Cas2 protein c. crRNA and Cas9 protein d. Cas1 and Cas2 proteins

Study smarter with the SolutionInn App


