Show calculation Show calculation Show calculations Show calculations Show calculations of percent error. of molality. of...
Fantastic news! We've Found the answer you've been seeking!
Question:

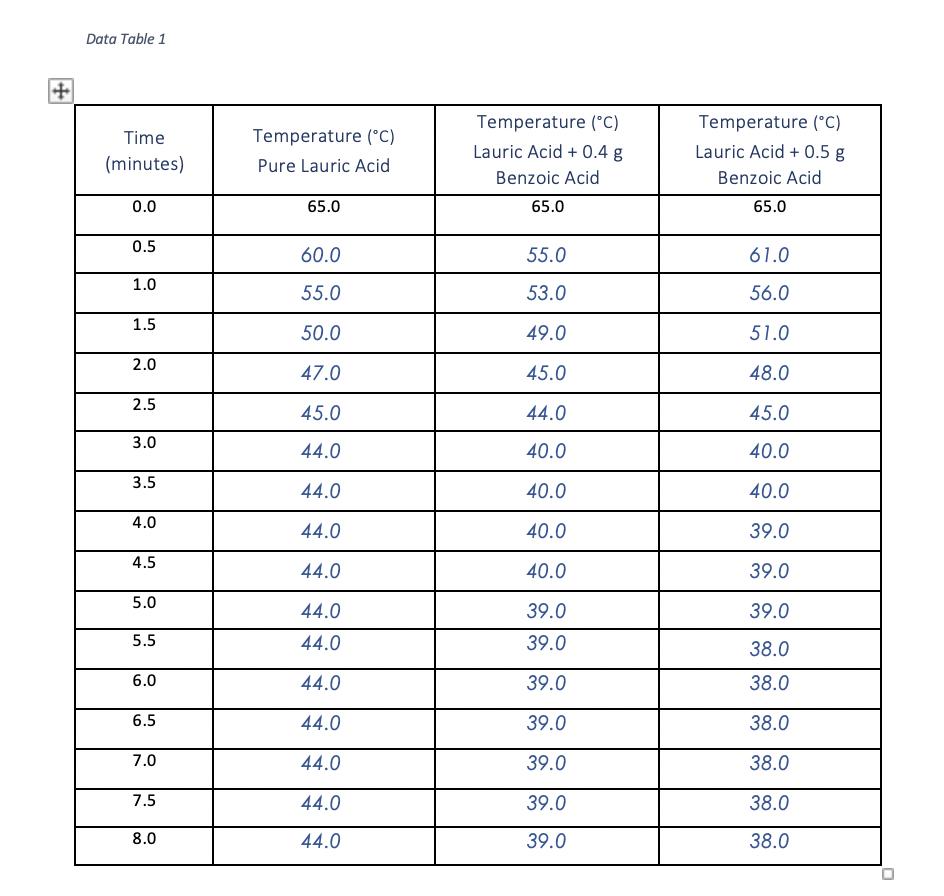
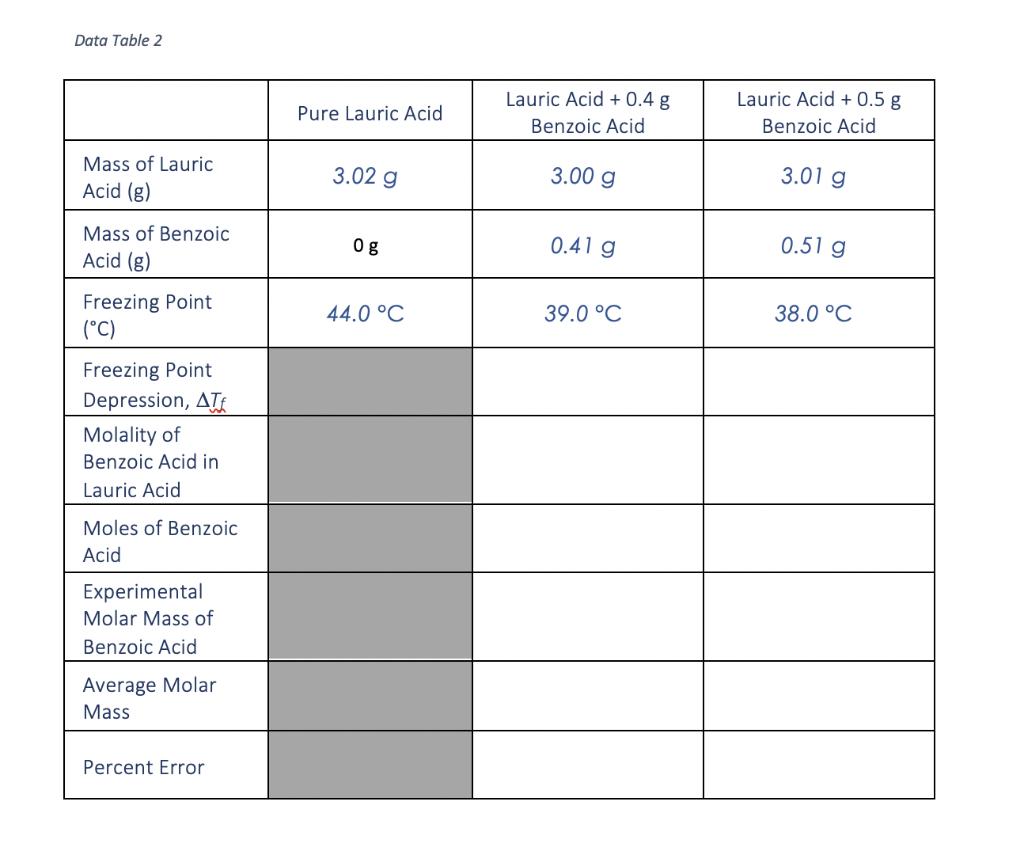
Transcribed Image Text:
Show calculation Show calculation Show calculations Show calculations Show calculations of percent error. of molality. of moles benzoic acid. of molar mass. of average molar mass. Data Table 1 Time (minutes) 0.0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 Temperature (°C) Pure Lauric Acid 65.0 60.0 55.0 50.0 47.0 45.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 Temperature (°C) Lauric Acid + 0.4 g Benzoic Acid 65.0 55.0 53.0 49.0 45.0 44.0 40.0 40.0 40.0 40.0 39.0 39.0 39.0 39.0 39.0 39.0 39.0 Temperature (°C) Lauric Acid + 0.5 g Benzoic Acid 65.0 61.0 56.0 51.0 48.0 45.0 40.0 40.0 39.0 39.0 39.0 38.0 38.0 38.0 38.0 38.0 38.0 U Data Table 2 Mass of Lauric Acid (g) Mass of Benzoic Acid (g) Freezing Point (°C) Freezing Point Depression, AT Molality of Benzoic Acid in Lauric Acid Moles of Benzoic Acid Experimental Molar Mass of Benzoic Acid Average Molar Mass Percent Error Pure Lauric Acid 3.02 g og 44.0 °C Lauric Acid + 0.4 g Benzoic Acid 3.00 g 0.41 g 39.0 °C Lauric Acid + 0.5 g Benzoic Acid 3.01 g 0.51 g 38.0 °C Show calculation Show calculation Show calculations Show calculations Show calculations of percent error. of molality. of moles benzoic acid. of molar mass. of average molar mass. Data Table 1 Time (minutes) 0.0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 Temperature (°C) Pure Lauric Acid 65.0 60.0 55.0 50.0 47.0 45.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 Temperature (°C) Lauric Acid + 0.4 g Benzoic Acid 65.0 55.0 53.0 49.0 45.0 44.0 40.0 40.0 40.0 40.0 39.0 39.0 39.0 39.0 39.0 39.0 39.0 Temperature (°C) Lauric Acid + 0.5 g Benzoic Acid 65.0 61.0 56.0 51.0 48.0 45.0 40.0 40.0 39.0 39.0 39.0 38.0 38.0 38.0 38.0 38.0 38.0 U Data Table 2 Mass of Lauric Acid (g) Mass of Benzoic Acid (g) Freezing Point (°C) Freezing Point Depression, AT Molality of Benzoic Acid in Lauric Acid Moles of Benzoic Acid Experimental Molar Mass of Benzoic Acid Average Molar Mass Percent Error Pure Lauric Acid 3.02 g og 44.0 °C Lauric Acid + 0.4 g Benzoic Acid 3.00 g 0.41 g 39.0 °C Lauric Acid + 0.5 g Benzoic Acid 3.01 g 0.51 g 38.0 °C
Expert Answer:
Answer rating: 100% (QA)
Calculate the freezing point depression T for each solution T Freezing Point of Pure Lauric Acid Fre... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these mathematics questions
-
1. What responsibilities might you have for ambulatory (Holter) monitoring? 2. Why is a diary a necessary part of Holter monitoring? 3. What does real-time monitoring mean? 4. Why should the patient...
-
Sometimes during ambulatory monitoring the electrodes may become loose or disconnected, and the patient will need to know how to handle this situation. Check the policy of the facility where you are...
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
The flexural rigidity of a rod-like structure like a microtubule is a measure of its bending stiffness. Flexural rigidity is given by EL, where E is the Young?s modulus and L is the geometrical...
-
Solve Prob. 4.138 assuming that the plumber exerts a force F= − (80 N)k and that the motor is turned off (M = 0). Problem 4.138: To clean the clogged drainpipe AE, a plumber disconnected both...
-
Find the total monthly payment, including taxes and insurance. Mortgage Interest Rate Term of Loan Annual Taxes Annual Insurance $79,500 4% 25 years $674 $228
-
There are two propositions: (a) I flip a coin. If it is heads, you are paid \$3; if it is tails, you are paid \(\$ 0\). It costs you \(\$ 1\) to participate in this proposition. You may do so at any...
-
Rhodes, Inc., is a fast-growing start-up firm that manufactures bicycles. The following income statement is available for July: Sales revenue (200 units @ $500 per unit) . . . . . . . $100,000 Less...
-
# Case Information: Aritzia Inc. The objective of this exercise is to develop your ability to perform a comprehensive analysis on a set of financial statements. Use the 2022 annual report of Aritzia...
-
In this mini-case you will perform some procedures required as a part of audit planning. For ease your audit manager has already organized the workpapers and completed several of the required...
-
Using Excel (please show all your formulas, calculations and work) Calculate the internal rate of return, net present value, payback period, profitability index and total increase in income after...
-
Explain the difference between intervening and superseding causes. a. Give an example of an intervening cause. b. Give an example of a superseding cause.
-
To prove negligence per se, the plaintiff must show that the defendant violated a statute and that the: a. statute was designed to protect against the kind of harm sustained by the plaintiff. b. the...
-
Professionals are held to a ____________ standard of care than those not in the profession.
-
True Or False Compliance with a statute does not necessarily establish that a defendant was negligent.
-
True Or False Under the majority rule, when a criminal statute is violated, courts do not apply the criminal statutory standard to civil cases.
-
Glen purchases a used church bus and plans to renovate it and use it as a camper. Glen rips out the seats, installs twin beds, builds a custom booth for eating and builds a countertop with a...
-
Use nodal analysis to determine voltages v1, v2, and v3 in the circuit Fig. 3.76. Figure 3.76 4 S 3i, 2 A 4A
-
A situation often encountered with thermodynamic data is that different sources may present different correlations for a given quantity. In this case study, for example, two different expressions are...
-
A liquid mixture containing 40.0 wt% n-octane and the balance n-decane flows into a tank mounted on a balance. The mass in kg indicated by the scale is plotted against time. The data fall on a...
-
The composition of a coal is determined by a proximate analysis. The coal is first finely ground and air-dried. Samples of the dried coal are then subjected to several operations, with the sample...
-
N = 230, n = 15, k = 200 Compute the mean and standard deviation of the hypergeometric random variable X.
-
N = 60, n = 8, k = 25 Compute the mean and standard deviation of the hypergeometric random variable X.
-
One study showed that in a certain year, airline fatalities occur at the rate of 0.011 deaths per 100 million miles. Find the probability that, during the next 100 million miles of flight, there will...

Study smarter with the SolutionInn App


