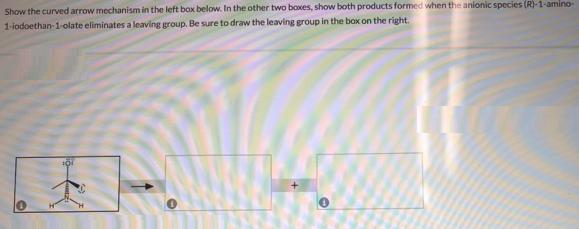
Show the curved arrow mechanism in the left box below. In the other two boxes, show...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Show the curved arrow mechanism in the left box below. In the other two boxes, show both products formed when the anionic species (R)-1-amino- 1-iodoethan-1-olate eliminates a leaving group. Be sure to draw the leaving group in the box on the right. Show the curved arrow mechanism in the left box below. In the other two boxes, show both products formed when the anionic species (R)-1-amino- 1-iodoethan-1-olate eliminates a leaving group. Be sure to draw the leaving group in the box on the right.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw curved arrows to show the flow of electrons responsible for the conversion of reactants into products. a. b. c. :O CH-C OCH
-
Two products are formed when methylenecyclohexane reacts with NBS. Explain how each is formed. CH2 CH Br CH2 NBS, peroxide
-
Draw both resonance structures of the enolate formed when each of the following ketones is treated with a strong base: (a) (b) (c) (d) (e)
-
HL Co. uses the high-low method to derive a total cost formula. Using a range of units produced from 1,500 to 7,500, and a range of total costs from $21,000 to $45,000, producing 2,000 units will...
-
In a control chart application, we have found that the grand average over all past samples of size 6 is x-bar = 30 and R-bar = 5. a. Set up x and R control charts for this application. b. The...
-
Most developed economies have well developed road and highway networks. From time to time new highways are built to relieve congested cities, but by and large most developed countries are not...
-
Redwing Floor Coverings, Inc., reported the following summarized data at December 31, 2009. Accounts appear in no particular order. Requirement 1. Prepare the trial balance of Redwing Floor Coverings...
-
Happy Times, Inc., wants to expand its party stores into the Southeast. In order to establish an immediate presence in the area, the company is considering the purchase of the privately held Joes...
-
Prove the following identity: cos(2x) - 1 sin(2x) tan(x) 1 cos(2x) - 1 sin(2x) Question Help: Video Submit Question Search ASUS VivoBook
-
SC Consulting, a supply chain consulting firm, must decide on the location of its home offices. Its clients are located primarily in the 16 states listed in Table 5-5. There are four potential sites...
-
Systems theory and market theory have captured much of modern organizational behavior.....What does systems theory say, and what does market theory of organization say? How do you relate it to your...
-
Use the linear wave equation to determine the wave speed of a wave modeled with the wave function y(x, t) = (0.10 m)sin[(1.50 m-1)x + (5.00 s)]. (Use the following as necessary: x and t. ay(x, t)...
-
Show that the following grammar is ambiguous. S - Ab I aaB A > a I Aa B -> b -
-
Oriole, Inc., has outstanding bonds that will mature in six years and pay an 8 percent coupon semiannually. If you paid $1,095.10 today and your required rate of return was 5.5 percent. (Round...
-
A solid sphere 20 cm in radius carries 19 C, distributed uniformly throughout its volume. LQ 4 Part A Find the electric field strength 10 cm from the sphere's center. Express your answer in...
-
One component of a metal sculpture consists of a solid uniform cube with an edge of length of 3 . 1 9 cm . The alloy used to make the cube has a density of 7 0 7 0 kg / m ^ 3 . What is the cubes mass
-
One The small town of Fort William has only two pharmacists. One pharmacist is Catholic, the other Protestant. side of the town is predominantly Catholic; the other predominantly Protestant. The...
-
In your readings, there were many examples given for nurturing close family relationships in this ever-evolving technological society we live in Based upon your readings and research describe three...
-
Determine the partial negative charge on the oxygen atom in a C=O bond. The bond length is 1.22 * and the bond dipole moment is 2.30 D.
-
Bupropion hydrochloride is an antidepressant marketed under the trade name Wellbutrin®. Propose a synthesis of bupropion hydrochloride, starting with benzene. Cl CCHCH3 NH2C(CH3)3CI bupropion...
-
The addition of HBr to which of the following alkenes is more highly regioselective? a. b. CH CH CH,CHC=CH, CH 3C CHCH or - CH CH3 or
-
6.17 Show that P
-
For a gas, if enthalpy is considered to be the function of temperature and pressure, then show that the Joule-Thomson coefficient is \[ \mu_{\mathrm{JT}}=-\frac{1}{C_{P}}\left(\frac{\partial...
-
What is inversion temperature? Mention the importance of this temperature in explaining the heating or cooling effect of a gas with the help of the Joule-Thomson inversion curve.

Study smarter with the SolutionInn App



