7.36. Sketch a temperature-composition phase diagram (see i Figures 7.14 and 7.15 for examples) for the...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
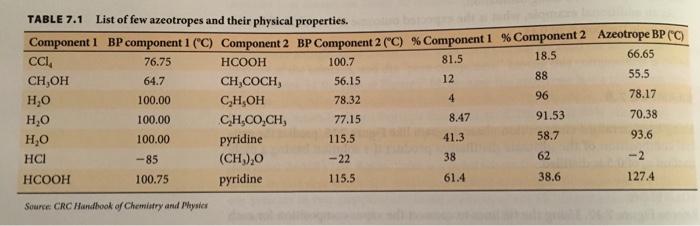
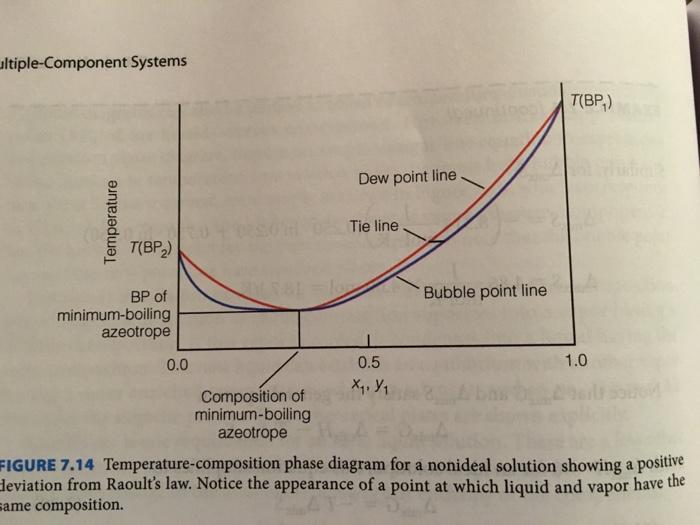
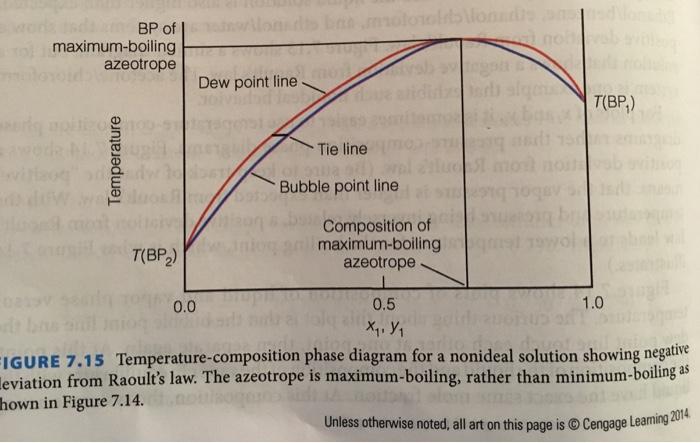
7.36. Sketch a temperature-composition phase diagram (see i Figures 7.14 and 7.15 for examples) for the CCl/HCOOH and HCOOH/pyridine solutions, using data from Table 7.1. How are the two phase diagrams different? How are they similar? TABLE 7.1 List of few azeotropes and their physical properties. Component 1 BP component 1 ("C) Component 2 BP Component 2 ("C) % Component 1 % Component 2 Azeotrope BP ("C) CC, 81.5 18.5 66.65 76.75 HCOOH 100.7 CH,OH 12 88 55,5 64.7 CH,COCH, 56.15 H,0 4 96 78.17 100.00 C,H,OH 78.32 100.00 CH,CO,CH, 8.47 91.53 70.38 77.15 H,O pyridine 115.5 41.3 58.7 93.6 100.00 HCI -85 (CH,),0 -22 38 62 -2 НСООН 100.75 pyridine 115.5 61.4 38.6 127.4 Source: CRC Handbook of Chemistry and Physies ultiple-Component Systems T(BP,) MAI Dew point line Tie line T(BP,) Bubble point line BP of minimum-boiling azeotrope 0.0 0.5 1.0 Composition of minimum-boiling azeotrope FIGURE 7.14 Temperature-composition phase diagram for a nonideal solution showing a deviation from Raoult's law. Notice the appearance of a point at which liquid and vapor have the same composition. positive Temperature Unless otherwise noted, all art on this page is © Cengage Leaming 2014 wodk sdam BP of | ilonsdts bammoloroltslonads dlon ol maximum-boiling noal azeotrope Dew point line adh alge T(BP,) Tie line lust pente Bubble point line br Composition of maximum-boiling azeotrope T(BP,) 0.0 0.5 1.0 X, Y, FIGURE 7.15 Temperature-composition phase diagram for a nonideal solution showing negative leviation from Raoult's law. The azeotrope is maximum-boiling, rather than minimum-boiling as hown in Figure 7.14. Temperature 7.36. Sketch a temperature-composition phase diagram (see i Figures 7.14 and 7.15 for examples) for the CCl/HCOOH and HCOOH/pyridine solutions, using data from Table 7.1. How are the two phase diagrams different? How are they similar? TABLE 7.1 List of few azeotropes and their physical properties. Component 1 BP component 1 ("C) Component 2 BP Component 2 ("C) % Component 1 % Component 2 Azeotrope BP ("C) CC, 81.5 18.5 66.65 76.75 HCOOH 100.7 CH,OH 12 88 55,5 64.7 CH,COCH, 56.15 H,0 4 96 78.17 100.00 C,H,OH 78.32 100.00 CH,CO,CH, 8.47 91.53 70.38 77.15 H,O pyridine 115.5 41.3 58.7 93.6 100.00 HCI -85 (CH,),0 -22 38 62 -2 НСООН 100.75 pyridine 115.5 61.4 38.6 127.4 Source: CRC Handbook of Chemistry and Physies ultiple-Component Systems T(BP,) MAI Dew point line Tie line T(BP,) Bubble point line BP of minimum-boiling azeotrope 0.0 0.5 1.0 Composition of minimum-boiling azeotrope FIGURE 7.14 Temperature-composition phase diagram for a nonideal solution showing a deviation from Raoult's law. Notice the appearance of a point at which liquid and vapor have the same composition. positive Temperature Unless otherwise noted, all art on this page is © Cengage Leaming 2014 wodk sdam BP of | ilonsdts bammoloroltslonads dlon ol maximum-boiling noal azeotrope Dew point line adh alge T(BP,) Tie line lust pente Bubble point line br Composition of maximum-boiling azeotrope T(BP,) 0.0 0.5 1.0 X, Y, FIGURE 7.15 Temperature-composition phase diagram for a nonideal solution showing negative leviation from Raoult's law. The azeotrope is maximum-boiling, rather than minimum-boiling as hown in Figure 7.14. Temperature
Expert Answer:
Answer rating: 100% (QA)
From the table we can see that CCl4HCOOH solution froms a minimum boiling azeotropes Hence it shows ... View the full answer

Related Book For 

Fundamentals of Investment Management
ISBN: 978-0078034626
10th edition
Authors: Geoffrey Hirt, Stanley Block
Posted Date:
Students also viewed these physics questions
-
Using data from Table 165 on page 426, assume you purchase a December 1100 (strike price) S&P 500 put option. Compute your total dollar profit or loss if the index has the following values at...
-
Using data from Table 8.4 on bond enthalpies, show that the more C---H bonds a molecule has compared to C-----O and O----H bonds, the more energy it can store.
-
Using data from Table 2.1 (p. 34), illustrate on the following graphs real GDP and population growth since 2000 (in the manner of Figure 2.1) for the nations indicated. China Canada Zimbabwe 100 100...
-
Given the project network that follows, compute the early, late, and slack times for the project. Be sure to show the early finish and late start times on yournetwork. 20 15 25 10 10 Slack IS B
-
When the U.S. Congress approved NAFTA, there was strong trade union and labor opposition. Do you agree with labor's opposition? Explain.
-
Gale Haley and Leah Manos formed a partnership, investing $180,000 and $60,000 respectively. Determine their participation in the year's net income of $150,000 under each of the following independent...
-
Jason Cannon purchased a defective tractor for which Cannon brought numerous causes of action against Bodensteiner Implement Company (Bodensteiner). One of the claims was breach of express...
-
Stagg Construction Co. is leasing equipment from Cloud Inc. The lease calls for payments of $50,000 a year plus $3,000 a year executory costs for five years. The first payment is due on January 1,...
-
1. A Pleasant Evening with Delta "functions" Let's define the Dirac delta "function" 8(x) by the property for "well-behaved" functions f. [ (x)6(x) dx = (0), (a) Consider the family of box functions....
-
Suppose that a learning algorithm is trying to find a consistent hypothesis when the classifications of examples are actually random. There are u Boolean attributes, and examples are drawn uniformly...
-
On January 1, 2001, sunshine corporation had 40000 shares of$10 par value common stock issued and outstanding. all 40,000shares had been issued in the prior period at $20 per share. On February 1,...
-
provide an example of Vocalics using verbal communication and in parenthesises explain how a non-verbal component of that message can contradict the verbal message ?
-
List applications or types of technology/mobile training programs that can be customized for differing employee development learning paths. Explain why these examples you listed can be effective.
-
Provide an example of Personal Presentation using Verbal Communication and then in parentheses explain how a non-verbal component of that message can contradict the verbal message ?
-
Provide an example of Haptics using Verbal Communication and then in parentheses explain how a non-verbal component of that message can contradict the verbal message ?
-
Record all employer's payroll expenses and liabilities for the month of July.
-
Make a Contact Us page as given in the following link : (HTML Doc) https://www.tune.com/contact-us/ You can skip the design part. However, the layout and content should be same. Do the Header and...
-
What are the three kinds of research types? Explain each type.
-
Compute the duration for the data in problem 1. Use an approach similar to that in Table 182 on page 469. A discount rate of 13 percent should be applied.
-
Explain the difference between a cash account and a margin account.
-
You have invested $1,000 in a 13 percent coupon bond that matures in five years. This bond is held in your individual retirement account, and you are not concerned about tax consequences. You are...
-
One difference between accounting for a governmental (not-for-profit) unit and a commercial (for-profit) enterprise is that a governmental unit should a. Not record depreciation expense in any of its...
-
Carson Citys general fund issued purchase orders of $630,000 to vendors for supplies. Which of the following entries should the city make to record this transaction? Debit Credit Q. ENCUMBRANCES...
-
Belle Valley incurred $100,000 of salaries and wages for the month ended March 31, 20X2. How should this be recorded on that date? c. Expenditures Salaries and Wages Debit Vouchers Payable Salaries...

Study smarter with the SolutionInn App


