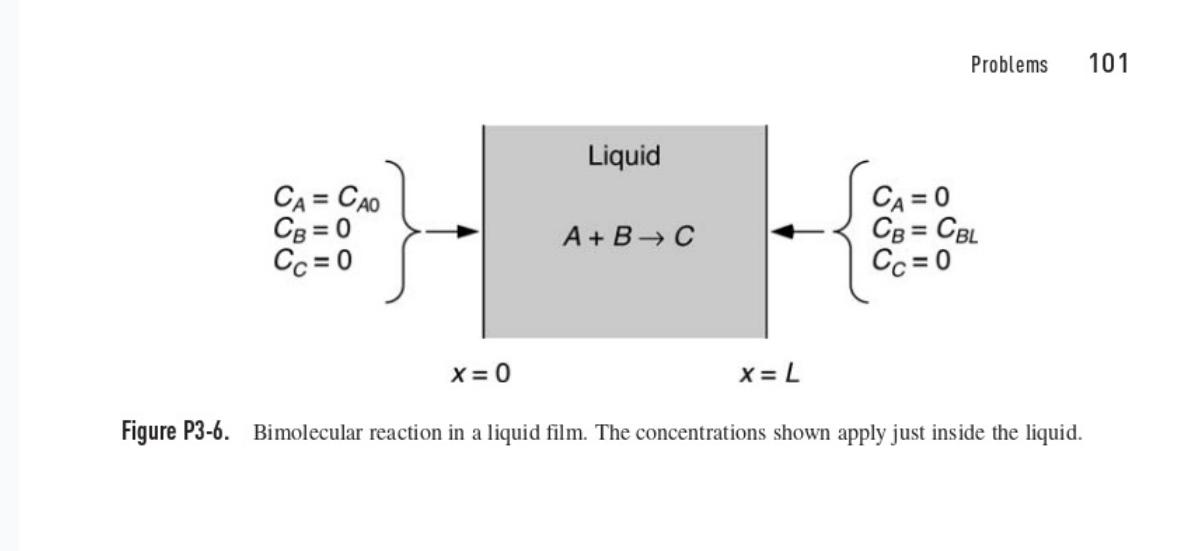
Slow Bimolecular Reaction This problem concerns the reaction A BC in a liquid film at steady state.
Question:
Slow Bimolecular Reaction This problem concerns the reaction A BC in a liquid film at steady state. The system is the same as in Problem 3-6, except that the reaction is assumed now to be relatively slow. As shown in Fig. P3-6, species A is introduced at x = 0 and species B at x = L. The bimolecular rate constant is k. For simplicity, assume that the diffusivities and boundary concentrations are equal, such that DA= DB = D and C po = CBL = Co. (a) Show that the concentration of A is governed by de £1(0 2n-1), 0(0) = 1, 8(1) = 0 dn? where 0 = C4/Co, n= x/L, and ε = Da = kCoL?/D. [Hint: First combine the conservation equations for A and B and solve for CB(x) in terms of CA(x).] (b) Use a perturbation analysis to determine (n) for slow reaction kinetics, for which <<< 1. Include terms through O(8²); that is, find the first three terms in the expansion.