Sodium nitrite, NaNO, has a molar mass of 85.0 g/mol. What volume of 1.60 M NaNO;...
Fantastic news! We've Found the answer you've been seeking!
Question:
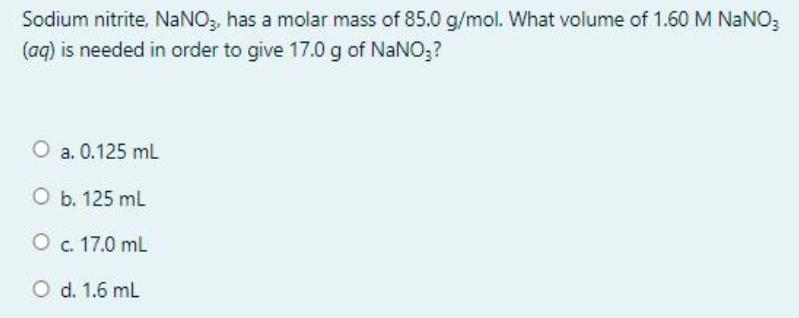
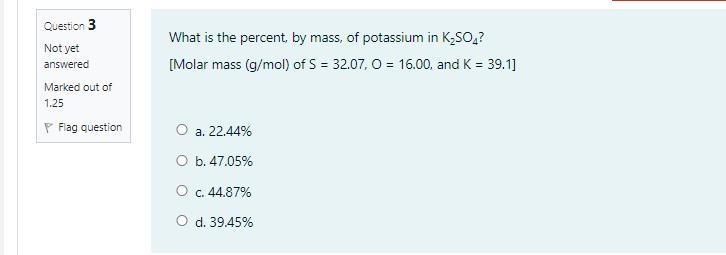

Transcribed Image Text:
Sodium nitrite, NaNO, has a molar mass of 85.0 g/mol. What volume of 1.60 M NaNO; (aq) is needed in order to give 17.0 g of NaNO;? O a. 0.125 mL O b. 125 mL O c. 17.0 ml O d. 1.6 mL Question 3 What is the percent, by mass, of potassium in K;SO,? Not yet [Molar mass (g/mol) of S = 32.07, O = 16.00, and K = 39.1] answered Marked out of 1.25 P Flag question O a. 22.44% а. O b. 47.05% O c. 44.87% O d. 39.45% Question 2 Given the thermochemical equation: 250,(g) + O2(g) – 2 so;(g) AH ren = -198 kJ How much heat is evolved when 192 g of SOz is burned? [molar mass (g/mol) of S = 32.00, and O = 16.00] Not yet answered Marked out of 1.25 P Flag question О а. 495 k O b. 297 kJ О с. 198 k O d. 396 kJ Sodium nitrite, NaNO, has a molar mass of 85.0 g/mol. What volume of 1.60 M NaNO; (aq) is needed in order to give 17.0 g of NaNO;? O a. 0.125 mL O b. 125 mL O c. 17.0 ml O d. 1.6 mL Question 3 What is the percent, by mass, of potassium in K;SO,? Not yet [Molar mass (g/mol) of S = 32.07, O = 16.00, and K = 39.1] answered Marked out of 1.25 P Flag question O a. 22.44% а. O b. 47.05% O c. 44.87% O d. 39.45% Question 2 Given the thermochemical equation: 250,(g) + O2(g) – 2 so;(g) AH ren = -198 kJ How much heat is evolved when 192 g of SOz is burned? [molar mass (g/mol) of S = 32.00, and O = 16.00] Not yet answered Marked out of 1.25 P Flag question О а. 495 k O b. 297 kJ О с. 198 k O d. 396 kJ
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A certain material has a molar mass of 20.0 g/mol, a Fermi energy of 5.00eV, and 2 valence electrons per atom. What is the density (g/cm3)?
-
A starch has a molar mass of 3.20 1004 g/mol. If 0.759 g of this starch is dissolved in 112 mL of solution, what is the osmotic pressure, in torr, at 25.00C?
-
(a) Oxygen (02) has a molar mass of 32.0 g/mol what is the average translational kinetic energy of an oxygen molecule at a temperature of 300 K? (b) What is the average value of the square of its...
-
Researchers examined forecasters' interest rate predictions for 34 quarters to see whether the predictions corresponded to what actually happened. The 2 x 2 contingency table below shows the...
-
As the first gift from their estate, Lily and Tom Phillips plan to give $20,000 to their son, Raoul, for a down payment on a house. a. How much gift tax will be owed by Lily and Tom? b. How much...
-
Paul adams own health club in downtown Los Angeles. He charges his customers an annual fee of $500 and has an existing customer base of 500. Paul plans to raise the annual fee by 6% every year and...
-
Do analytical efforts, big data examinations, and textual analyses impact compliance and fraud deterrence? Provide an explanation for your opinion.
-
1. Consider Figure Between points c and d, the opportunity cost of ___ tons of wheat is _______ tons of steel. 2. Arrow up or down: An increase in the wage for high-school graduates ______ the...
-
The relation below concerns Invoice information. INVOICE (INV-No, INV-DATE, CUSTOMER-No, CUST-NAME, CUST-ADDRESS, SWPACK-ID, SWPACK-DESCR, UNIT-PRICE, SWPACK-QTY) For a given invoice (identified by...
-
Determine the monthly cash flows and total cash generated at the end of each month and just before the payment is received for the construction of a house with the following budget and schedule. On...
-
what is the linking verb and complement in the sentences below? The caller could have been Jeremy. What is the linking verb? What is the complement? It was he who called yesterday. What is the...
-
Shad Corporation, Meshach Inc, and Abed Co., will each pay a dividend of $3.65 in the coming year. The required rate of return for their stock is 8%, 11% and 14% respectively. In addition, they each...
-
Classic Car Dealers has an average of 150 cars in inventory. They sold 3000 cars this past year Assuming 365 days in a work year , how many days worth of inventory do they carry on average?
-
lowmo Ltd . is a private company that currently prepares its consolidated Sfinancial statements in accordance with ASPE. But since it has plans to go public in the next 2 - 3 years, it is considering...
-
Explain how the strategy will add value to shareholders who are interested in profitability, earnings per share, and future growth. You do not need to produce financial statements. You only need to...
-
Total shareholders' equity $1,959,993 $1,777,502 Net earnings for the year ended December 31, 2015, is $367,530. WestJet reported the following information in the notes to its 2015 financial...
-
A careful analysis of demand for Bubbles in Strasburg, ND reveals a strange segmentation in the market. (RecallBubbles is the beverage which produces an unexplained craving for Lawrance Welk's music....
-
The Dow Jones Industrial Average reached a high of $ 7801.63 on December 29, 1997. Recall from Example 18.4 that it reached a high of $ 1003 on November 14, 1972. The Consumer Price Index for...
-
Natural gas consists primarily of methane, CH4. It is used in a process called steam reforming to prepare a gaseous mixture of carbon monoxide and hydrogen for industrial use. CH4(g) + H2O(g) CO(g)...
-
Which of the following particles has the longest wavelength? a. An electron traveling at x meters per second b. A proton traveling at x meters per second c. A proton traveling at 2x meters per second
-
Give the names of the following ions. a. Mn2+ b. Ni2+ c. Co2+ d. Co3+
-
A free-trade equilibrium exists in which the United States exports food and imports clothing. U.S. engineers now invent a new process for producing clothing at a lower cost. This process cannot be...
-
A number of Latin American countries export coffee and import other goods. A longterm drought now reduces coffee production in the countries of this region. Assume that they remain exporters of...
-
Developing a new exportable natural resource can cause problems. One, discussed later in this chapter, is the problem of immiserizing growth: If you are already exporting and your export expansion...

Study smarter with the SolutionInn App


