Starting with propene show a multi step synthesis of 1,4 pentadiene. Show a multi step synthesis...
Fantastic news! We've Found the answer you've been seeking!
Question:
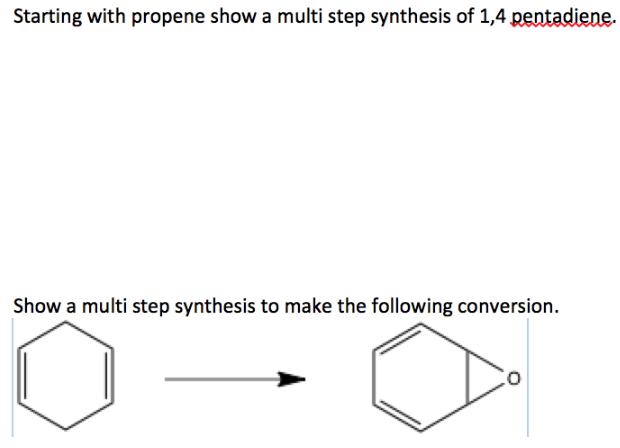
Transcribed Image Text:
Starting with propene show a multi step synthesis of 1,4 pentadiene. Show a multi step synthesis to make the following conversion. Starting with propene show a multi step synthesis of 1,4 pentadiene. Show a multi step synthesis to make the following conversion.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The Pentire Corporation is looking to make the following annual dividend payments over the next four years, commencing one year from today: $10, $14, $7 and $2 respectively. After this time (four...
-
The dietitian wants your department to make the following recipe for lunch service in the Assisted Living Facility in which you work, where there are 125 residents. For this exercise, we will not...
-
Ray Solutions decided to make the following changes in its accounting policies on January 1, 2011: 1. Changed from the cash to the accrual basis of accounting for recognizing revenue on its service...
-
The two roots of a quadratic equation ax 2 + bx + c = 0 can be obtained using the following formula: b 2 - 4ac is called the discriminant of the quadratic equation. If it is positive, the equation...
-
1. Describe whether your organization is exploitive, impoverished, supportive, or enlightened. 2. Describe the features of your organization that indicate which kind of organization it is. 3....
-
Determine the natural frequencies and mode shapes of the system of Figure P7.19 if \(k=12,000 \mathrm{~N} / \mathrm{m}\) and each bar is of mass \(12 \mathrm{~kg}\) and length \(4 \mathrm{~m}\). x1...
-
a. Zero bending moment b. Zero transverse displacement c. Zero shear force d. Zero slope \(W^{\prime \prime}=0\)
-
Debbie Dean issued a check to Betty Brown payable to the order of Cathy Cain and Betty Brown. Betty indorsed the check Payable to Elizabeth East, (signed) Betty Brown. What rights, if any, does...
-
Provide examples of disintermediation, reintermediation & info mediation models. Provide an example of an online store whose webpage design is consistent with 7C's. What technologies can be used to...
-
1. What is your assessment of the financial performance of Nelson Nurseries? 2. Do you agree with Christine Barton?s accounts-payable policy? 3. What explains the erosion of the cash balance? 4. What...
-
Use the following information and complete the form: Schedule 8812 John Doe is the chief financial officer of Facebook, located in New York, NY. John has a 10 year old son, Phil, who lives in his...
-
What is a Balanced Scorecard? Of the four perspectives that this concept introduces, do you think one of the perspectives is more crucial than another? Does it depend on the company in particular?...
-
The maximum consequence for a willful violation that could result in a worker's death carries a penalty of seventy thousand dollars. Jeffress mentions companies see OSHA as a mosquito. How I see this...
-
Given the following budgets, what would be prepared first when completing the master budget? Answer The direct materials budget The cash budget The pro-forma income statement The manufacturing...
-
A company seeking a line of credit at a bank was turned down. Among other things, the bank stated that the company's 2 to 1 current ratio was not adequate. Give reasons why a 2 to 1 current ratio...
-
Examine the Summary Compensation table. What types of compensation does the firm grant its executives? Based on the descriptions of the forms of compensation in the Compensation Discussion and...
-
A compact object of mass m is in a close orbit around a luminous supergiant star of mass M. Mass flows off of the supergiant and accretes onto the compact object at a rate that does not change the...
-
Derive Eq. (18.33) from Eq. (18.32).
-
Predict the products formed by sodium hydroxide-promoted dehydrohalogenation of the following compounds. In each case, predict which will be the major product. (a) 1-bromobutane (b) 2-chlorobutane...
-
Urylon fibers are used in premium fishing nets because the polymer is relatively stable to UV light and aqueous acid and base. The structure of Urylon is (a) What functional group is contained in the...
-
Show how crossed Claisen condensations could be used to prepare the following esters. (a) (b) (c) (d) Ph C-CH-C OCH,CH CH CH-C-OCH3 C-C-oCH Eto C CH-C OCH,CH Ph CH,CH,CH,
-
The internal energy of an ideal gas depends on (a) Temperature, specific heat and volume (b) Temperature and specific heat (c) Temperature, specific heat and pressure (d) Pressure, volume and...
-
In the ammonia synthesis reaction \(\mathrm{N}_{2}+3 \mathrm{H}_{2}=2 \mathrm{NH}_{3}+22.4 \mathrm{kcal}\), the formation of \(\mathrm{NH}_{3}\) will be favoured by (a) High temperature (b) Low...
-
At constant temperature, the volume of a given mass of a gas is inversely proportional to the pressure. This is known as (a) Avogadro's law (b) Charles' law (c) Boyle's law (d) none of these.

Study smarter with the SolutionInn App



