8. 8 pts. The setup of an artificial kidney dialysis system is shown below. Assuming the...
Fantastic news! We've Found the answer you've been seeking!
Question:
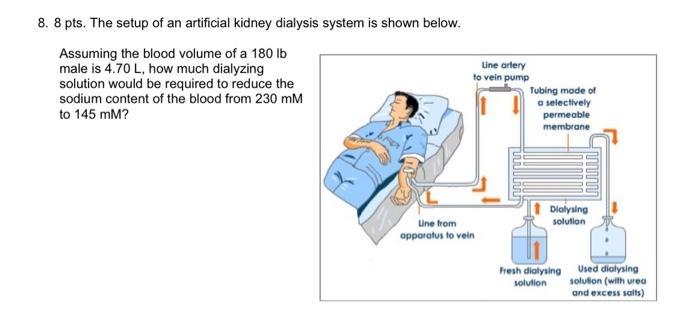

Transcribed Image Text:
8. 8 pts. The setup of an artificial kidney dialysis system is shown below. Assuming the blood volume of a 180 lb male is 4.70 L, how much dialyzing solution would be required to reduce the sodium content of the blood from 230 mM to 145 mm? Line artery to vein pump Line from apparatus to vein Tubing made of a selectively permeable membrane Dialysing solution Fresh dialysing solution Used dialysing solution (with urea and excess salts) 4. The amino acid to the right would exist as drawn: a. Below pH 2.2 b. Between pH 2.2 and 6.00 c. Between pH 6.00 and pH 9.4 d. Above pH 9.4 e. The amino acid shown would not exist at any pH HN- 5. Provide the charge of the following polypeptide at pH 8.5: DFYWRPA a. -2 b. -1 c. 0 d. +1 e. The correct answer isn't listed a. acetic acid, carbonic acid, nitrous acid, phosphoric acid b. carbonic acid, acetic acid, nitrous acid, phosphoric acid c. acetic acid, nitrous acid, carbonic acid, phosphoric acid d. phosphoric acid, nitrous acid, acetic acid, carbonic acid e. carbonic acid, phosphoric acid, nitrous acid, acetic acid ⒸNH3 6. List the acids in INCREASING order of strength (weakest to strongest): nitrous acid (Ka = 4.0 × 104) carbonic acid (Ka = 4.4 x 10-7) acetic acid (Ka = 1.7 x 10-5) phosphoric acid (Ka = 7.3 × 10-³) 8. 8 pts. The setup of an artificial kidney dialysis system is shown below. Assuming the blood volume of a 180 lb male is 4.70 L, how much dialyzing solution would be required to reduce the sodium content of the blood from 230 mM to 145 mm? Line artery to vein pump Line from apparatus to vein Tubing made of a selectively permeable membrane Dialysing solution Fresh dialysing solution Used dialysing solution (with urea and excess salts) 4. The amino acid to the right would exist as drawn: a. Below pH 2.2 b. Between pH 2.2 and 6.00 c. Between pH 6.00 and pH 9.4 d. Above pH 9.4 e. The amino acid shown would not exist at any pH HN- 5. Provide the charge of the following polypeptide at pH 8.5: DFYWRPA a. -2 b. -1 c. 0 d. +1 e. The correct answer isn't listed a. acetic acid, carbonic acid, nitrous acid, phosphoric acid b. carbonic acid, acetic acid, nitrous acid, phosphoric acid c. acetic acid, nitrous acid, carbonic acid, phosphoric acid d. phosphoric acid, nitrous acid, acetic acid, carbonic acid e. carbonic acid, phosphoric acid, nitrous acid, acetic acid ⒸNH3 6. List the acids in INCREASING order of strength (weakest to strongest): nitrous acid (Ka = 4.0 × 104) carbonic acid (Ka = 4.4 x 10-7) acetic acid (Ka = 1.7 x 10-5) phosphoric acid (Ka = 7.3 × 10-³)
Expert Answer:
Answer rating: 100% (QA)
8 DIALYSIS The separation of particles in a liquid on the ba... View the full answer

Related Book For 

Contemporary Business Mathematics with Canadian Applications
ISBN: 978-0134141084
11th edition
Authors: S. A. Hummelbrunner, Kelly Halliday, Ali R. Hassanlou, K. Suzanne Coombs
Posted Date:
Students also viewed these biology questions
-
Thirsty Limited's business involves the bottling and distribution of a wide variety of carbonated soft drinks. Some drinks are developed internally, whilst other brands are purchased. The following...
-
If money is worth 6% compounded annually, what amount today is equivalent to $10,000 paid: a. 12 years from now? b. 24 years from now? c. 36 years from now?
-
If money is worth 8% compounded quarterly, how long (to the nearest day) before a scheduled payment of $6000 will $5000 be an equivalent payment? For the purpose of determining the number of days in...
-
In Figure 3-4, the current position of the demand curve is D 1 , and the price of a wireless earbud, which is a normal good, is $3. If there is an increase in consumer incomes, will the demand curve...
-
What is the difference between performance management and performance appraisal?
-
AstroStar, Inc., has a board of directors consisting of three members (Eckhart, Dolan, and Macero) and has approximately five hundred shareholders. At a regular board meeting, the board selects...
-
Parents of minors took Apple to court in 2012 for supplying game applications, on iPhones, that were free but through which users could purchase in-game currencies. Apparently, parents would log on...
-
(Transaction AnalysisService Company) Beverly Crusher is a licensed CPA. During the first month of operations of her business (a sole proprietorship), the following events and transactions occurred....
-
Consider the following logarithmic equation. log3(x) log.x 8- log(x) log(x) = 1. (i) Find the value(s) of a satisfying in the equation. (ii) Determine for what values of the logarithmic expression on...
-
1. How will you characterize Tupperwares distribution strategy in relation to the theoretical models? 2. What are the advantages and disadvantages of Tupperwares distribution model? 3. How do you...
-
Find vector field of {(1,0), (1, 1), (1,-1), (-1,0), (-1,-1)] For transformation f(x, y) = (1,2)
-
discuss in detail a life cycle analysis related to The Cancer Care Center at the University of Colorado Anschutz. You may also use this online article for additional information: Basic Overview of...
-
What is the trading statistics of both firms: In preparing and Ratio Analysis for both Coca-Cola and PepsiCo what is the Trading Statistics of both the Firms, Coca-Cola and PepsiCo.
-
Your project deadline is Nov-30, which requires 10-days to complete. Based on the duration, you need to start the project no later than Nov-20. You are available to start the project as soon as...
-
Discuss how you would secure and manage supplier contracts by completing the following activities: Explain how you would utilise contract and relationship management techniques, tools and...
-
How to find the market rent of a commercial property with a 25-year lease, with 5-year rent reviews , at an initial rent of $76,000 per annum at a yield of 4%
-
A high-speed photograph of a club hitting a golf ball is shown in the figure below. Ted Kinsman/Photo Researchers, Inc. The club was in contact with a ball, initially at rest, for about 0.0017 s. If...
-
Graph the following conic sections, labeling vertices, foci, directrices, and asymptotes (if they exist). Give the eccentricity of the curve. Use a graphing utility to check your work. 10 5 + 2 cos 0
-
A seven-month note dated November 1, 2019, earning interest at 7.5%, has a maturity value of $6000. Find the face value of the note.
-
On a trip, a motorist purchased gasoline as follows: 56 litres at $0.99 per litre; 64 litres at $1.065 per litre; 70 litres at $1.015 per litre; and 54 litres at $1.045 per litre. (a) What was the...
-
Sport Alive sold 72 pairs of ski poles. Superlight poles sell at $30 per pair, while ordinary poles sell at $16 per pair. If the total sales value was $1530, how many pairs of each type were sold?
-
Many hotel jobs are inherently dead end; for example, maids, laundry workers, and valets, either have no great aspirations to move up, or are just using these jobs temporarily, for instance, to help...
-
Using what you learned in this chapter of Dessler Human Resource Management, build on the companys new system by recommending two more specific career development activities the hotel should...
-
Should other employees (cleaner/spotters, counter people) be put on a similar plan? Why or why not? If so, how exactly?

Study smarter with the SolutionInn App


