Sunland Corporation had the following comparative current assets and current liabilities: Cash Current assets Short-term investments...
Fantastic news! We've Found the answer you've been seeking!
Question:

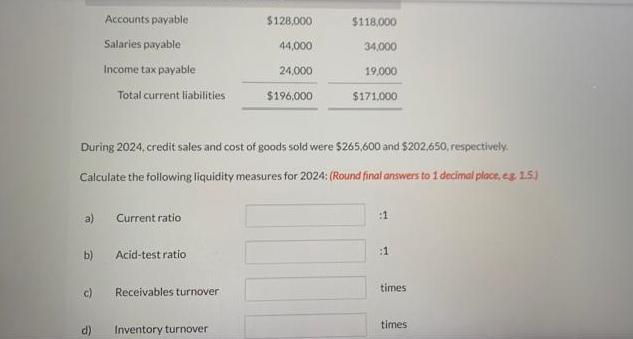
Transcribed Image Text:
Sunland Corporation had the following comparative current assets and current liabilities: Cash Current assets Short-term investments Accounts receivable Inventory Prepaid assets Dec. 31, 2024 Total current assets $37,960 48,000 63,000 106,000 39,040 $294,000 Current liabilities Dec. 31, 2023 $38,000 18,000 103,000 87,000 28,000 $274,000 a) b) (c) Accounts payable Salaries payable Income tax payable d) Total current liabilities During 2024, credit sales and cost of goods sold were $265,600 and $202,650, respectively. Calculate the following liquidity measures for 2024: (Round final answers to 1 decimal place, eg. 15) Current ratio Acid-test ratio Receivables turnover $128,000 44,000 24,000 $196,000 Inventory turnover $118,000 34,000 19,000 $171.000 :1 :1 times times Sunland Corporation had the following comparative current assets and current liabilities: Cash Current assets Short-term investments Accounts receivable Inventory Prepaid assets Dec. 31, 2024 Total current assets $37,960 48,000 63,000 106,000 39,040 $294,000 Current liabilities Dec. 31, 2023 $38,000 18,000 103,000 87,000 28,000 $274,000 a) b) (c) Accounts payable Salaries payable Income tax payable d) Total current liabilities During 2024, credit sales and cost of goods sold were $265,600 and $202,650, respectively. Calculate the following liquidity measures for 2024: (Round final answers to 1 decimal place, eg. 15) Current ratio Acid-test ratio Receivables turnover $128,000 44,000 24,000 $196,000 Inventory turnover $118,000 34,000 19,000 $171.000 :1 :1 times times
Expert Answer:
Answer rating: 100% (QA)
Answer a Current Ratio Current Assets cReceivable turnover Net credit sal... View the full answer

Related Book For 

Financial Accounting and Reporting a Global Perspective
ISBN: 978-1408076866
4th edition
Authors: Michel Lebas, Herve Stolowy, Yuan Ding
Posted Date:
Students also viewed these accounting questions
-
The following is the balance sheet and income statement for YUM, Inc. (the company that operates Kentucky Fried Chicken and Pizza Hut), in condensed form, plus some information from the cash flow...
-
Walton Corporation had the following comparative current assets and current liabilities: During 2017, net credit sales and cost of goods sold were $475,000 and $250,000, respectively. Net cash...
-
Theo is depositing $1,300 today in an account with an expected rate of return of 8.1 percent. If he deposits an additional $3,200 two years from today, and $4,000 three years from today, what will...
-
Sarah works for an accounting company. She has a lot of administrative procedures to follow and little flexibility in the way she performs her daily work. Sarah's job has a high level of _____....
-
Given a flow network G = (V, E), let f1 and f2 be functions from V V to R. The flow sum f1 + f2 is the function from V V to R defined by (26.4) (fi + f2) (u, v) = f1 (u, v) + f2(u, v) for all u, v ...
-
A piston, mp= 5 kg, is fitted in a cylinder, A = 15 cm2, that contains a gas. The setup is in a centrifuge that creates an acceleration of 25 m/s2 in the direction of piston motion towards the gas....
-
When would you use a motion to strike?
-
Rob Davis, Stewart Vintu, and Vern Wilson are liquidating their partnership. Before selling the assets and paying the liabilities, the capital balances are Davis $40,000; Vintu, $24,000; and Wilson,...
-
Give an example of two securities, one issued by a financial institution and another held as an asset by the same financial institution, that reflect intermediation.
-
Go through the transaction list (Document1) and identify which cycle each transaction belongs to. A transaction will belong to only one cycle; either the Sale to Cash Receipt (S/CR) Cycle, Purchases...
-
Gender socialization is the process of learning the social expectations and attitudes associated with one's sex. Sociologists explain through gender socialization why human males and females behave...
-
Completely factor the expression. 5x2 17x 24 5 96 48
-
Evaluate the principles of crystallization in microgravity environments, considering how reduced gravitational forces impact nucleation, growth, and aggregation, and discussing the potential for...
-
You are a correctional counselor assigned to James, a 17-year-old boy who was charged with robbery after he held up a cab driver while holding a knife to his throat. James was soon apprehended by...
-
Despite a court ruling against them, a Florida couple vows to continue their legal battle to gain custody of a child born by the woman they hired as a surrogate, but who then decided to keep the...
-
Indicate the effect, if any, that each separate transaction has on financing cash flows. Note: Select "No Effect" if there is no effect. a. Long-term notes payable with a carrying value of $15,000...
-
At a service centre customers arrive at the rate of 10 per hour and are served at the rate of 15 per hour. Their arrival follows poisson distribution and service in exponentially distributed. Find...
-
Write a program to move a signed number from smaller register to bigger register. Hint: movzx ax, bl Topic: Data Related Operators and Directives in assembly language
-
Saint Gobain, a France-based global enterprise, is involved in the production and sales of flat glass and building materials. It reports in its financial statements for the year ended 31 December...
-
Bosch is a German group producing automotive equipment, power tools and home appliances. The consolidated balance sheet and notes to financial statements 2010 and 2011 show the following elements...
-
Below are excerpts from Notes to consolidated financial statements of four companies reporting movements in provisions. Stora Enso (Finland IFRS Source: Annual report 2011 Production of paper)...
-
The heat of vaporization of ether is \(25.98 \mathrm{~kJ} / \mathrm{mol}\) at its boiling point, \(34.5^{\circ} \mathrm{C}\). (a) Calculate the rate of change of vapour pressure with temperature...
-
Show that the internal energy of an ideal gas is a function only of temperature.
-
For a gas obeying the equation of state \(V=B+\frac{R T}{P}\), the Joule-Thomson coefficient is given by \[ \mu_{\mathrm{JT}}=-\frac{1}{C_{P}}\left(T \frac{d B}{d T}-B ight) \]

Study smarter with the SolutionInn App


