Suppose that 1.5 mole of CH4 and 2 moles of O2 are burned. The resultant products include:
Fantastic news! We've Found the answer you've been seeking!
Question:
Suppose that 1.5 mole of CH4 and 2 moles of O2 are burned. The resultant products include: 0.60 mole CO2 0.40 mole CO How many moles of CH4, O2 and H2O are in the products?Reactions:
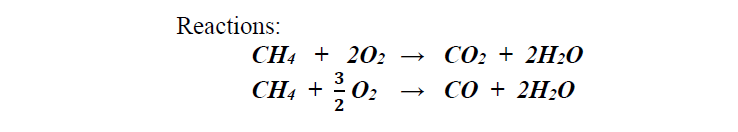
Related Book For 

Posted Date:




