Suppose you have decided to buy flashlights for your car. You decided to purchase for 20,100...
Fantastic news! We've Found the answer you've been seeking!
Question:
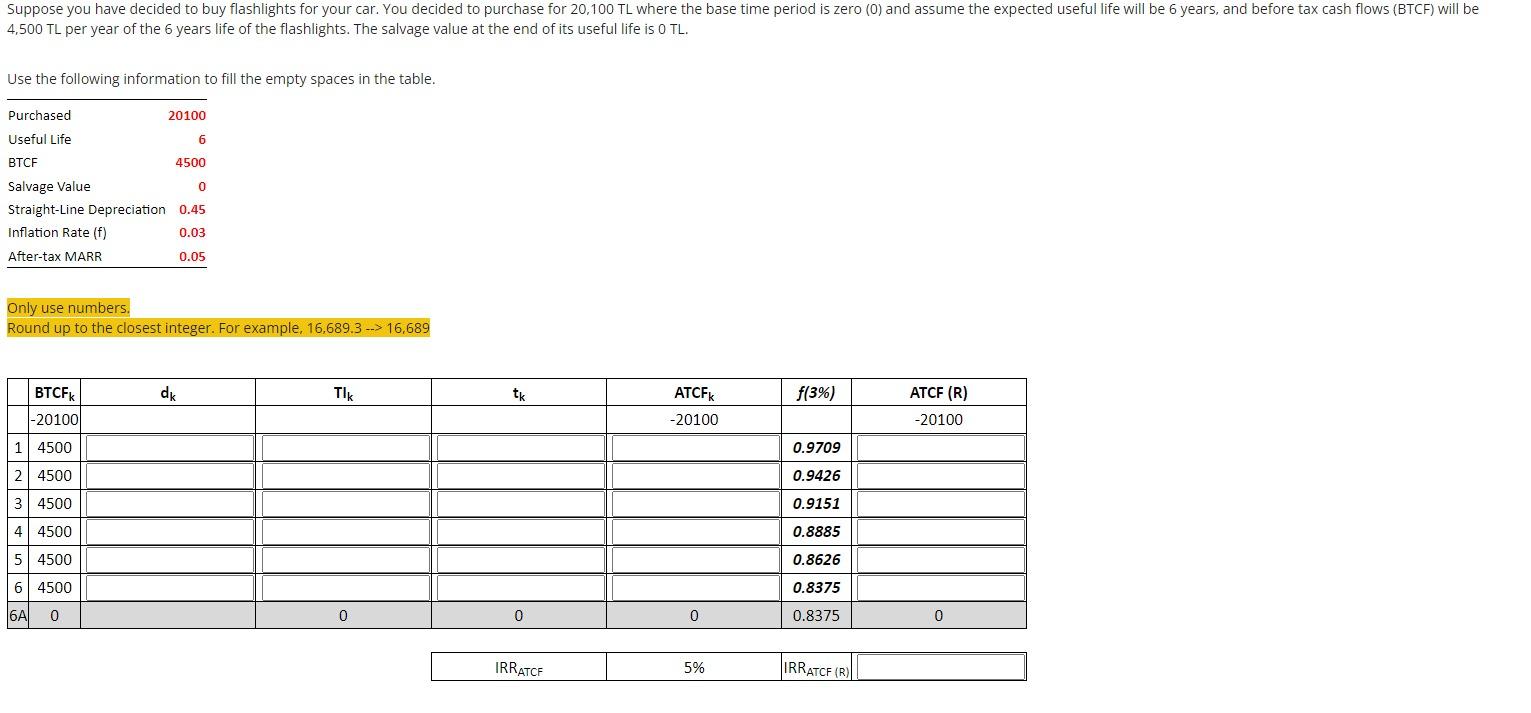
Transcribed Image Text:
Suppose you have decided to buy flashlights for your car. You decided to purchase for 20,100 TL where the base time period is zero (0) and assume the expected useful life will be 6 years, and before tax cash flows (BTCF) will be 4.500 TL per year of the 6 years life of the flashlights. The salvage value at the end of its useful life is 0 TL. Use the following information to fill the empty spaces in the table. Purchased Useful Life BTCF Salvage Value Straight-Line Depreciation Inflation Rate (f) After-tax MARR BTCFK -20100 20100 1 4500 2 4500 3 4500 4 4500 5 4500 6 4500 6A 0 6 4500 Only use numbers. Round up to the closest integer. For example, 16.689.3 16,689 0 0.45 0.03 0.05 dk TIK 0 tk 0 IRRATCE ATCFK -20100 0 5% f(3%) 0.9709 0.9426 0.9151 0.8885 0.8626 0.8375 0.8375 IRRATCF (R) ATCF (R) -20100 0 Suppose you have decided to buy flashlights for your car. You decided to purchase for 20,100 TL where the base time period is zero (0) and assume the expected useful life will be 6 years, and before tax cash flows (BTCF) will be 4.500 TL per year of the 6 years life of the flashlights. The salvage value at the end of its useful life is 0 TL. Use the following information to fill the empty spaces in the table. Purchased Useful Life BTCF Salvage Value Straight-Line Depreciation Inflation Rate (f) After-tax MARR BTCFK -20100 20100 1 4500 2 4500 3 4500 4 4500 5 4500 6 4500 6A 0 6 4500 Only use numbers. Round up to the closest integer. For example, 16.689.3 16,689 0 0.45 0.03 0.05 dk TIK 0 tk 0 IRRATCE ATCFK -20100 0 5% f(3%) 0.9709 0.9426 0.9151 0.8885 0.8626 0.8375 0.8375 IRRATCF (R) ATCF (R) -20100 0
Expert Answer:
Answer rating: 100% (QA)
The provided image contains a table to be filled out for the financial analysis of purchasing flashlights for a car The table includes columns for beforetax cash flow BTCFk depreciation dk aftertax ca... View the full answer

Related Book For 

Advanced Financial Accounting
ISBN: 978-0137030385
6th edition
Authors: Thomas Beechy, Umashanker Trivedi, Kenneth MacAulay
Posted Date:
Students also viewed these economics questions
-
Jereld Kim founded Kimco LTD, a firm that is currently one of the largest producers of paper and pulp with annual sales of AUD 800 Million. Kim located his first plant in a rural town in Tasmania,...
-
Suppose you were just told that you will receive an end-of-the-year bonus of $15,000 from your company. Suppose further that your marginal income tax rate is 33.33% which means that you will have to...
-
Large Mart has previously attempted to develop a study pillow which would have allowed students to upload study material into their brains whilst sleeping. However, Large Mart has recently discovered...
-
Find the limit L. Then use the - definition to prove that the limit is L. lim x4 x
-
At week 24 of a project to shoot a television commercial, what should the expenditures be? If the earned value is right on schedule but the actual expenses are $9,000, what are the cost and schedule...
-
Compute the rotational KE of a 25-kg wheel rotating at 6.0 rev/s if the radius of gyration of the wheel is 22 cm.
-
Aaron Reed, a photographer, was in a dispute with Ezelle Investment Properties, Inc., over Ezelle allegedly using one of Reeds photographs without permission. Reed sent Ezelle a cease-and-desist...
-
You are provided with the following information for Ramirez Enterprises, effective as of its April 30, 2014, year-end. Accounts payable............ $ 834 Accounts receivable............ 810...
-
Explain the relationship and the difference between online analytical processing systems and customer relationship management systems within a business intelligence program.?
-
La-Z-Boy Incorporated included the following information in its 2009 annual report (dollars in millions). Define solvency and discuss how this information might be useful in assessing the company??s...
-
The pH scale measures how acidic or basic a substance is. pH is defined as the negative logarithm of the hydrogen ion activity in an aqueous solution, a H . Thus, if a H = 0.01, then pH = -log 0.01 =...
-
What Are Your Arguments? Why Should We Care Construct at least 4 different claims (four different "I Says") supporting your working thesis statement, which could in turn be supported by evidence from...
-
3. Using the table below, which displays information for the same perfectly competitive firm during the short-run, answer the following questions. Units of Labor, L 234 5 6 7 Total Product (TP) 20 30...
-
Discussion question it doses not have to be long (minimum 250 words and, at least, one source current) Textbook Fitzsimmons, J., Fitzsimmons, M., & Bordoloi, S. (2014). Service management:...
-
Explain the main characteristics of an active equity investment strategy based on market timing. [10 marks] II. Consider the following information about the performance of two fund managers during...
-
Linux disk management includes several important tasks such as adding or removing storage devices, creating and deleting partitions, mounting partitions on appropriate directories and making file...
-
calculate the molarity of the solution. the volume of the standard solution is 250 ml. The molar mass of potassium iodate is 214.001 g mol ^-1. Mass of KIO3 0.630 g M= ((mass KlO3 (g)/ molar mass...
-
Show that, given a maximum flow in a network with m edges, a minimum cut of N can be computed in O(m) time.
-
On January 1, 20X0, Mariachi Corporation acquired 70% of the outstanding shares of Sombrero Company for $ 85,000 cash. On that date, Sombrero Company had $ 35,000 of common shares outstanding and $...
-
How does the accounting exposure under the temporal method differ from the accounting exposure under the current-rate method?
-
What is negative goodwill? How should it be reported in the consolidated statements?
-
Write a MATLAB program to compute the CA-CFAR threshold value. Use a similar approach to that used in the case of a fixed threshold.
-
Starting with Eq. (13.81), show that as \(\mathrm{N}\) is increased so is the over all probability of false alarm. More specifically, prove that \(P_{F A} \approx N P_{f a}\). Equation (13.81) PFA...
-
Design a cylindrical RCS calibration target such that its broadside RCS (cylinder) and end (flat plate) RCS are equal to 10 m2 at f = 9.5 GHz. The RCS for a flat plate of area A is fp = 4f2A2/c2.

Study smarter with the SolutionInn App


