Susquehanna Valley Limousine has modified their automobile engine to provide heat for an oven to bake...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
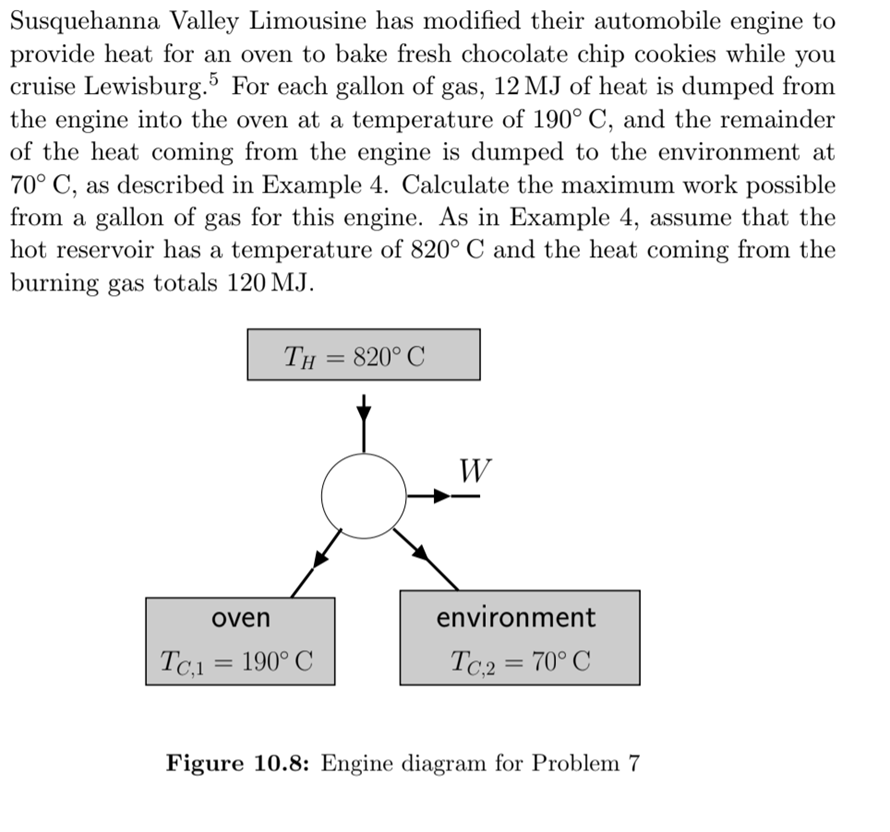
Susquehanna Valley Limousine has modified their automobile engine to provide heat for an oven to bake fresh chocolate chip cookies while you cruise Lewisburg.5 For each gallon of gas, 12 MJ of heat is dumped from the engine into the oven at a temperature of 190° C, and the remainder of the heat coming from the engine is dumped to the environment at 70° C, as described in Example 4. Calculate the maximum work possible from a gallon of gas for this engine. As in Example 4, assume that the hot reservoir has a temperature of 820° C and the heat coming from the burning gas totals 120 MJ. Th = 820° C W oven environment Tc.1 = 190° C Tc,2= 70° C Figure 10.8: Engine diagram for Problem 7 Susquehanna Valley Limousine has modified their automobile engine to provide heat for an oven to bake fresh chocolate chip cookies while you cruise Lewisburg.5 For each gallon of gas, 12 MJ of heat is dumped from the engine into the oven at a temperature of 190° C, and the remainder of the heat coming from the engine is dumped to the environment at 70° C, as described in Example 4. Calculate the maximum work possible from a gallon of gas for this engine. As in Example 4, assume that the hot reservoir has a temperature of 820° C and the heat coming from the burning gas totals 120 MJ. Th = 820° C W oven environment Tc.1 = 190° C Tc,2= 70° C Figure 10.8: Engine diagram for Problem 7
Expert Answer:
Answer rating: 100% (QA)
Solution THEO200 120mJ for reversible process 12myy then oven Q1 03 Tc19090 ... View the full answer

Related Book For 

Financial and Managerial Accounting
ISBN: 978-1133940593
10th edition
Authors: Belverd E. Needles, Marian Powers, Susan V. Crosson
Posted Date:
Students also viewed these accounting questions
-
Consider a daily lottery as described in Example 5.5.4. a. Compute the probability that two particular days in a row will both have triples. b. Suppose that we observe triples on a particular day....
-
Determine the access function f(aij), as described in Example 5.10(d), for a matrix A = {alj)m n, where (a) m = 12, n = 12; (b) m = 7, n = 10; (c) m = 10, n = 7.
-
Consider a random walk as described in Example 9.13. After one million steps, find the probability that the walk is within 500 steps from the origin.
-
Consider the following grooves, each of width W, that have been machined from a solid block of material. (a) For each case obtain an expression for the view factor of the groove with respect to the...
-
You are the lender of last resort and an institution approaches you for a loan. You assess that the institution has $800 million in assets, mostly in long-term loans, and $600 million in liabilities....
-
The function g(x) = x 2 + 3px + (14p 3), where p is an integer, has two equal roots. a. Find the value of p. b. For this value of p, solve the equation x 2 + 3px + (14p 3) = 0.
-
What is a System Storage Concept? Identify examples of its UCs.
-
State whether the following provisions in a note impair or preclude negotiability, the instrument in each instance being otherwise in proper form. Answer each statement with either the word...
-
Given the following free cash flows. determine the IRR for the three independent projects A, B, and C. Data Table Initial outlay (Click on the following icon in order to copy its contents into a...
-
The following summarized data (amounts in millions) are taken from the September 26, 2020, and September 28, 2019, comparative financial statements of Apple Incorporated, a company that designs,...
-
A small 42 g ball strikes the ground at a speed of 22 m/s. It rebounds from the surface at a speed of 17 m/s after being in contact for 3.1 ms. I. Approximate Evaluation First, we'll look at this...
-
When creating their newest product, Seagram's Escapes, Seagram's wanted to expand to a new market segment who wanted to enjoy light alcoholic drinks, and not get drunk quickly. They were hoping to...
-
1-Write a project paper on any company of your choice considering all the variables mentioned in the question herein. The first part of the presentation should provide a) a brief background of the t...
-
Describe your ideal team. Where could I find a suitable person for that role? Who in your personal and professional network can you contact for help finding candidates?
-
Ministry of Health wants to locate a new referral hospital in any one of the following locations: - Likoni, Changamwe or Kisauni. Develop four Factors for your evaluation and advice where to locate...
-
discuss and analyse the key cultural and environmental issues which could influence a selected company's approach to digital marketing in the selected country. One should address the impact on...
-
Two workers are pushing and pulling a box. The force F1 is 1000N at 45 from the x-axis and the force F2 is 500 N, 30 up from the x-axis. (a) Replace these two forces by an equivalent system of a...
-
Describe a group you belong or have belonged discuss the stages of group development and suggest how to improve the group effectiveness by using the group development model.
-
Prepare journal entries to record the transactions for Matt Ing's sole proprietorship that follow. 1. Ing invests $5,000 in his business. 2. Ing closed his Income Summary account's credit balance of...
-
Selected accounts from Dence's Gourmet Shop's adjusted trial balance as of March 31, 2014, the end of the current fiscal year, follow. The merchandise inventory for Dence's was $81,222 at the...
-
Jennifer Polk opened a school for administrative skills called Polk Office Training, Inc., and completed the following transactions: a. Contributed the following assets to the business in exchange...
-
The hydrodynamic boundary layer equations consist of one momentum equation and the continuity equation. How does one solve for the pressure?
-
How does the hydrodynamic boundary layer thickness depend upon the Reynolds number?
-
If the liquid has a high thermal conductivity, the heat transfer coefficient depends on \(\mathrm{Pr}^{1 / 2}\). What is the root cause of the increase dependence on \(\mathrm{Pr}\) ?

Study smarter with the SolutionInn App


