Symbol Name 17C 13C 22Ne 345 Ca Kr 94Zr carbon-12 carbon-13 neon-22 neon-21 sulfur-34 chlorine-37 krypton-82...
Fantastic news! We've Found the answer you've been seeking!
Question:
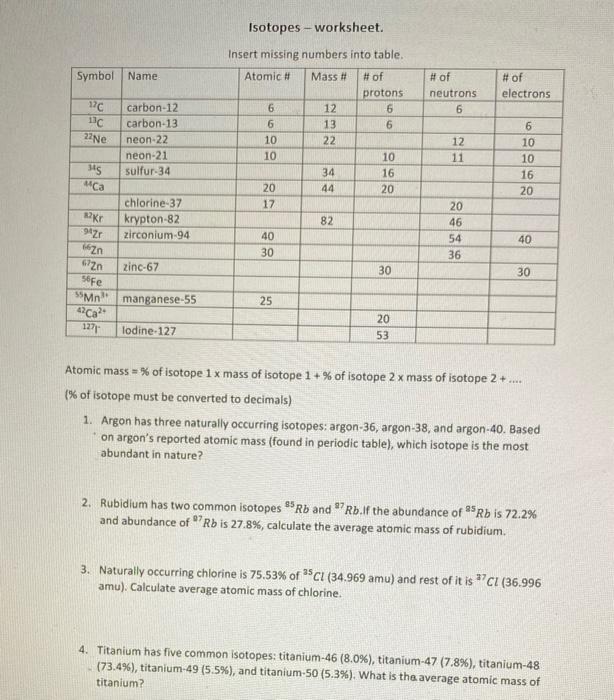
Transcribed Image Text:
Symbol Name 17C 13C 22Ne 345 Ca Kr 94Zr carbon-12 carbon-13 neon-22 neon-21 sulfur-34 chlorine-37 krypton-82 zirconium-94 Zn 672n 56Fe 55Mn 42Ca2+ 1271 lodine-127 zinc-67 manganese-55 Isotopes - worksheet. Insert missing numbers into table. Atomic # Mass # # of protons 6 6 6 10 10 20 17 40 30 25 232 12 13 34 44 82 6 10 16 20 30 20 53 # of neutrons 6 12 11 20 46 54 36 # of electrons 6 10 10 16 20 40 30 Atomic mass=% of isotope 1 x mass of isotope 1+% of isotope 2 x mass of isotope 2+.... (% of isotope must be converted to decimals) 1. Argon has three naturally occurring isotopes: argon-36, argon-38, and argon-40. Based on argon's reported atomic mass (found in periodic table), which isotope is the most abundant in nature? 2. Rubidium has two common isotopes 85Rb and 7Rb.If the abundance of 25 Rb is 72.2% and abundance of 7 Rb is 27.8%, calculate the average atomic mass of rubidium. 3. Naturally occurring chlorine is 75.53% of 35Cl (34.969 amu) and rest of it is 7 Cl (36.996 amu). Calculate average atomic mass of chlorine. 4. Titanium has five common isotopes: titanium-46 (8.0%), titanium-47 (7.8%), titanium-48 (73.4%), titanium-49 (5.5%), and titanium-50 (5.3%). What is the average atomic mass of titanium? Symbol Name 17C 13C 22Ne 345 Ca Kr 94Zr carbon-12 carbon-13 neon-22 neon-21 sulfur-34 chlorine-37 krypton-82 zirconium-94 Zn 672n 56Fe 55Mn 42Ca2+ 1271 lodine-127 zinc-67 manganese-55 Isotopes - worksheet. Insert missing numbers into table. Atomic # Mass # # of protons 6 6 6 10 10 20 17 40 30 25 232 12 13 34 44 82 6 10 16 20 30 20 53 # of neutrons 6 12 11 20 46 54 36 # of electrons 6 10 10 16 20 40 30 Atomic mass=% of isotope 1 x mass of isotope 1+% of isotope 2 x mass of isotope 2+.... (% of isotope must be converted to decimals) 1. Argon has three naturally occurring isotopes: argon-36, argon-38, and argon-40. Based on argon's reported atomic mass (found in periodic table), which isotope is the most abundant in nature? 2. Rubidium has two common isotopes 85Rb and 7Rb.If the abundance of 25 Rb is 72.2% and abundance of 7 Rb is 27.8%, calculate the average atomic mass of rubidium. 3. Naturally occurring chlorine is 75.53% of 35Cl (34.969 amu) and rest of it is 7 Cl (36.996 amu). Calculate average atomic mass of chlorine. 4. Titanium has five common isotopes: titanium-46 (8.0%), titanium-47 (7.8%), titanium-48 (73.4%), titanium-49 (5.5%), and titanium-50 (5.3%). What is the average atomic mass of titanium?
Expert Answer:
Answer rating: 100% (QA)
Answer Symbol Name 17C 13C 22Ne 21 Ne 345 MCa 37 CI 82Kr 94Zr 66Zn 672n 56FC 55Mn 42Ca2 1271 carbon1... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The element oxygen has three naturally occurring isotopes, with 8, 9, and 10 neutrons in the nucleus, respectively. (a) Write the full chemical symbols for these three isotopes. (b) Describe the...
-
Of the isomeric C5H11+ carbocations, which one is the most stable?
-
Which currency is the most widely preferred currency of exchange in global financial markets? Why?
-
What is path dependence? Explain the concept using your own words and provide an example of path dependence (other than what we have already mentioned in class). List the 4 categories of innovation,...
-
Kane Inc.'s bank statement from Western Bank at August 31, 2010, gives the following information. Balance, August 1 $16,400 Bank debit memorandum: A summary of the Cash account in the ledger for...
-
Why do you think results are so mixed on the use of social networking in the workplace? What are some possible upsides to utilizing social media as part of an integrated marketing strategy,...
-
Nancy Carpenter Optical Dispensary purchased supplies on account for \($1,200\). Two weeks later, the business paid half on account. Requirements 1. Journalize the two transactions for Nancy...
-
Ghose Company manufactures basketballs. Materials are added at the beginning of the production process and conversion costs are incurred uniformly. Production and cost data for the month of July 2011...
-
1.Basis of Islamic Accounting Theory The history of the birth of Islamic accounting is inseparable from the development of Islam, the obligation to record non-cash transactions (see QS. Al-Baqarah:...
-
A company is intending to invest in a capital budgeting project to manufacture a medical testing device and has projected the following sales: Year1 Year2 Year3 Year4 Year5 50,000 66,400 ...
-
The electric field is constant over each face of the cube shown in (Figure 1). Figure 10- 10 5 10 < 2018 W2 2019 ndf 1 of 1 20 15 Field strengths in N/C Copyright 2019 Pearson Education Inc. Al >...
-
Mega Sdn Bhd is a local producer and seller of an infrared device used to assist vehicles' driver in paying the tolls known as SmartBAG. This product is highly demanded in the market because it...
-
b. If the query point q moves, the resulting q.3NN can change, and the point where q.3NN changes is called the safe exit point. Find the same exit point in the figure above when q moves in n2...
-
7. Solve the modified diffusion equation, taking into account heat generation in the bar: =a2+ +uo sin x, (0,1) 10, (L,t) = 30, = y(x, 0) = f(x), 0 0 0
-
Show that the projection into the -plane of the curve of intersection of the parabolic cylinder =132 and the paraboloid =2+2 is an ellipse. Find a vector-parametric equation for the curve of...
-
A group of people are asked to target weighing out 75 lbs for each sample. There were 78 people who participated in this study. The next page shows a control chart of their measurements. The average...
-
7 Taxpayer acquired a building on November 30, 2021. During December, 2021, the taxpayer incurs $100,000 for routine repairs including, $20,000 for fixing plumbing leaks, $50,000 for painting, and...
-
Write each fraction as a percent. 7 50
-
A 2.17-g sample of barium reacted completely with water. What is the equation for the reaction? How many milliliters of dry H 2 evolved at 21C and 748 mmHg?
-
An electron is accelerated through a potential difference of 15.6 kilovolts (giving the electron a kinetic energy of 15.6 keV). What is the associated wavelength of the electron in angstroms?
-
Chloroform, CHCl 3 , is a solvent and has been used as an anesthetic. The heat of vaporization of chloroform at its boiling point (61.2C) is 29.6 kJ/mol. What is the entropy change when 5.10 mol CHCl...
-
When sampling without replacement, if the sample size is less than ______________ % of the population, the sampled items may be treated as independent. In Exercises 710, fill in each blank with the...
-
A jar contains 4 red marbles, 3 blue marbles, and 5 green marbles. Two marbles are drawn from the jar one at a time without replacement. What is the probability that the second marble is red, given...
-
A compound event is formed by combining two or more events. In Exercises 912, determine whether the statement is true or false. If the statement is false, rewrite it as a true statement.

Study smarter with the SolutionInn App


