Isotopes of an element have the same number of protons in thenucleus, but different numbers of neutrons.
Fantastic news! We've Found the answer you've been seeking!
Question:
Isotopes of an element have the same number of protons in thenucleus, but different numbers of neutrons. A magnetic field can beused to separate different isotopes. Consider singly ionizedpositive ions of three isotopes of the same element; they all havethe same net charge, but their masses are different. As they enterthe magnetic field all have the same velocity. However, they followdifferent paths through the magnetic field -labeled I, II and III inthe diagram. Complete the following statement by selecting thecorrect path
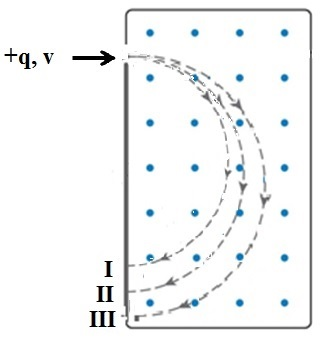
The isotope having the greatest mass follows _________ throughthe magnetic field.
Related Book For 

Posted Date:




