T=800 C- a Si T Xmax a + 1 1/2 X = Xp P GS...
Fantastic news! We've Found the answer you've been seeking!
Question:
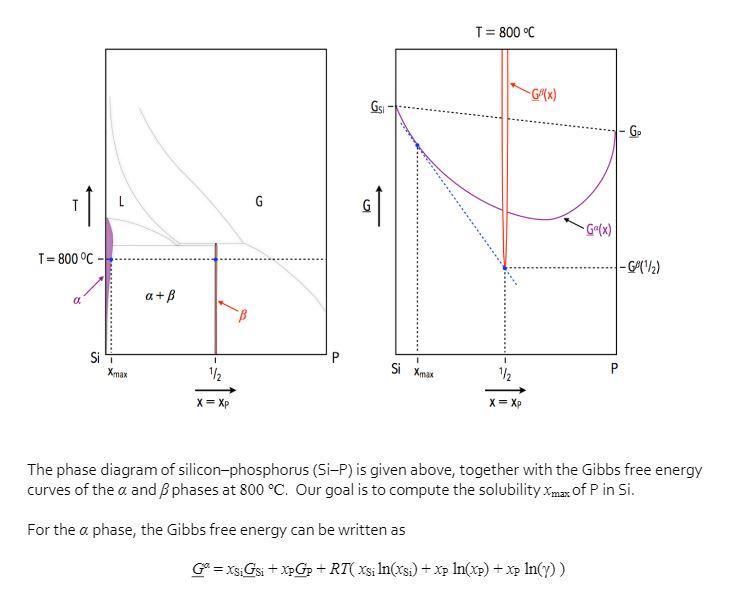

Transcribed Image Text:
T=800 °C- a Si T Xmax a + ß 1 1/2 X = Xp P GS I Si Xmax T = 800 °C 1/₂ X = Xp -G²(x) -Ga(x) G = XsiGsi + XpGp + RT(xsi ln(xsi) + xp ln(xp) + xp ln(y)) P Go -Gº (¹/2) The phase diagram of silicon-phosphorus (Si-P) is given above, together with the Gibbs free energy curves of the a and 3 phases at 800 °C. Our goal is to compute the solubility Xmax of P in Si. For the a phase, the Gibbs free energy can be written as where y is a constant equal to y = ¹2. For the phase, the Gibbs free energy is very sharp around xp = ¹/2 and its minimum value is GB(¹/2) = ¹/2Gsi +¹/2Gp + AG where AG = -20,600 J/mol. (1) Give the expression of the chemical potential of Si in the a phase usi in terms of Gsi and xsi? Show your derivation. (2) Give the expression of the chemical potential of P in the a phase up in terms of Gp, xp, and y? Show your derivation. (3) By examining the Gibbs free energy curves, it is seen that the tangent to Gºat x = xmax must intersect with the tip of G. From this observation, show that Xxmax must fulfill 2AG = RT In(max(1-Xmax)). (4) Since the solubility is expected to be small, it is safe to make the approximation Xmax(1-Xmax)~Xmax Using this approximation, calculate the solubility Xmax (y=¹/2, AG = -20,600 J/mol, T = 800 °C) T=800 °C- a Si T Xmax a + ß 1 1/2 X = Xp P GS I Si Xmax T = 800 °C 1/₂ X = Xp -G²(x) -Ga(x) G = XsiGsi + XpGp + RT(xsi ln(xsi) + xp ln(xp) + xp ln(y)) P Go -Gº (¹/2) The phase diagram of silicon-phosphorus (Si-P) is given above, together with the Gibbs free energy curves of the a and 3 phases at 800 °C. Our goal is to compute the solubility Xmax of P in Si. For the a phase, the Gibbs free energy can be written as where y is a constant equal to y = ¹2. For the phase, the Gibbs free energy is very sharp around xp = ¹/2 and its minimum value is GB(¹/2) = ¹/2Gsi +¹/2Gp + AG where AG = -20,600 J/mol. (1) Give the expression of the chemical potential of Si in the a phase usi in terms of Gsi and xsi? Show your derivation. (2) Give the expression of the chemical potential of P in the a phase up in terms of Gp, xp, and y? Show your derivation. (3) By examining the Gibbs free energy curves, it is seen that the tangent to Gºat x = xmax must intersect with the tip of G. From this observation, show that Xxmax must fulfill 2AG = RT In(max(1-Xmax)). (4) Since the solubility is expected to be small, it is safe to make the approximation Xmax(1-Xmax)~Xmax Using this approximation, calculate the solubility Xmax (y=¹/2, AG = -20,600 J/mol, T = 800 °C)
Expert Answer:
Answer rating: 100% (QA)
1 The chemical potential of Si in the a phase si can be derived from the expression of Gibbs free energy as follows G12 12GSi 12Gp AG Since G12 represents the minimum value of the Gibbs free energy fo... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The interval (2, 5) can be written as the inequality _________.
-
A linearly polarized wave can be written as the superposition of two oppositely circularly polarized waves that have equal amplitudes, show that for the case considered. the plane of linear...
-
The phase diagram of a hypothetical substance is (a) Estimate the normal boiling point and freezing point of the substance. (b) What is the physical state of the substance under the following...
-
Draw the array's dimensions and contents of the following initialized two-dimensional arrays: 1) 2) int[][] arr1= new int [3] [4]; arr1 [0] [1]-5; arrl [0] [2] = 10: arrl [1] [2]-7; arri [0] [3] = 9;...
-
(a) Cody Company sells three different categories of tools (small, medium, and large). The cost and market value of its inventory of tools are as follows Determine the value of the companys inventory...
-
New Look Vision Group Inc. manufactures and retails eye care products. The company operates company stores under different banners including New Look Eyewear, Vogue Optical, Iris, and Greiche &...
-
Compare and discuss the estimated longitudinal compressive strengths of Scotchply 1002 E-glass/epoxy based on (a) fiber microbuckling and (b) transverse tensile rupture. Assume linear elastic...
-
Omega Chemicals Limited took a $ 420,000 two- year note receivable from a customer in connection with a major sale transaction on 1 May 20X7. The note required annual 31 April interest payments of...
-
Do you think that people under the age of 18 should be required to wear protective helmets when skateboarding, in-line, bicycling, snowboarding, or skiing? Why or why not.
-
A large roll of paper having a mass of 20 kg and a radius r = 150 mm is resting over the edge of a corner, such that the end of the paper on the roll is attached to the horizontal surface. If the...
-
Required: a. Prepare the income statement for the year ended 31 December 2021. b. Prepare the Statement of Financial Position as at 31 December 2021.
-
General Electric formerly entered a foreign market by either acquiring an established firm or establishing a greenfield subsidiary. Joint ventures with a local company were almost never considered....
-
A shock to aggregate supply will also have different outcomes when there are different assumptions about the formation of the level of expected inflation. As in Question 4, one assumption is that the...
-
IBMs CEO, Sam Palmisano, likes to talk about the evolution of global strategy at one of the worlds largest computer enterprises. According to Palmisano, when IBM first started to expand...
-
Convergence in two sets of countries Consider three rich countries: France, Belgium, and Italy, and four poor countries, Ethiopia, Kenya, Nigeria, and Uganda. Define for each country the ratio of its...
-
Back in 1970, companies in the United States assembled more than 15 million bicycles a year. Then globalization took hold. As cross-border tariffs tumbled, U.S. bicycle companies increasingly...
-
a) b) Based on Table 1, draw a diagram to show the distributed database environment scenario. Table CAMPUS STUDENT STAFF ELEARNING Table 1: DDBMS Environment Fragments CAMPUS 1 CAMPUS 2 N/A N/A N/A...
-
Organizations are increasing their use of personality tests to screen job applicants. What are some of the advantages and disadvantages of this approach? What can managers do to avoid some of the...
-
Show that for air at 20C the speed of a tube in 1ftes per second is given by, approximately. where the length L and diameter d are in centimeters; we have tried to correct for end effects on tube of...
-
The lower 10-15 km of the atmosphere-the troposphere-is often in a convective steady state at constant entropy, not constant temperature. In such equilibrium pVy is independent of altitude, where =...
-
Calculate the temperature of the surface of the Earth on the assumption that as a black body in thermal equilibrium it reradiates as much thermal radiation as it receive from the Sun. Assume also...
-
A food processor claims that at most \(10 \%\) of her jars of instant coffee contain less coffee than claimed on the label. To test this claim, 16 jars of her instant coffee are randomly selected and...
-
Refer to Exercise 4.2. (a) Determine the cumulative probability distribution \(F(x)\). (b) Graph the probability distribution of \(f(x)\) as a bar chart and below it graph \(F(x)\). Data From...
-
Four emergency radios are available for rescue workers but one does not work properly. Two randomly selected radios are taken on a rescue mission. Let \(X\) be the number that work properly between...

Study smarter with the SolutionInn App


