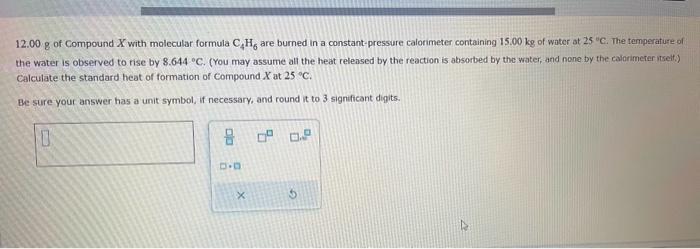
12.00 g of Compound X with molecular formula CH, are burned in a constant-pressure calorimeter containing...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
12.00 g of Compound X with molecular formula C₂H, are burned in a constant-pressure calorimeter containing 15.00 kg of water at 25 °C. The temperature of the water is observed to rise by 8.644 °C. (You may assume all the heat released by the reaction is absorbed by the water, and none by the calorimeter itself.) Calculate the standard heat of formation of Compound X at 25 °C. Be sure your answer has a unit symbol, if necessary, and round it to 3 significant digits. 0 8 D.D X 0.² 0.2 5 12.00 g of Compound X with molecular formula C₂H, are burned in a constant-pressure calorimeter containing 15.00 kg of water at 25 °C. The temperature of the water is observed to rise by 8.644 °C. (You may assume all the heat released by the reaction is absorbed by the water, and none by the calorimeter itself.) Calculate the standard heat of formation of Compound X at 25 °C. Be sure your answer has a unit symbol, if necessary, and round it to 3 significant digits. 0 8 D.D X 0.² 0.2 5
Expert Answer:
Answer rating: 100% (QA)
Here heat released in reaction is absorved by water So Heat gained by water 9 wa... View the full answer

Related Book For 

Accounting Principles
ISBN: 978-1118875056
12th edition
Authors: Jerry Weygandt, Paul Kimmel, Donald Kieso
Posted Date:
Students also viewed these accounting questions
-
Tabares Corporation had these transactions during 2014. (a) Issued $50,000 par value common stock for cash. (b) Purchased a machine for $30,000, giving a long-term note in exchange. (c) Issued...
-
Tabares Corporation had these transactions during 2017. (a) Issued $50,000 par value common stock for cash. (b) Purchased a machine for $30,000, giving a long-term note in exchange. (c) Issued...
-
Tabares Corporation had these transactions during 2019. (a) Issued $50,000 par value common stock for cash. (b) Purchased a machine for $30,000, giving a long-term note in exchange. (c) Issued...
-
You require inventory and accounts receivable collateral for all C&I loans. You have a guideline of an advance rate of 70% for customer receivables of less than 60 days of age. Older receivables get...
-
Consider a household that uses 14,000 kWh of electricity per year and 900 gallons of fuel oil during a heating season. The average amount of CO2 produced is 26.4 lbm/ gallon of fuel oil and 1.54...
-
Wilma purchased a used minivan, including a three-month warranty on parts and labour, from Xenon Used Cars Inc. for $10 000. She paid $1500 down and signed a promissory note for $8500, payable in...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically attractive?...
-
Exercise 18.11 is an extension of Exercise 18.10. The following information pertains to May activity in Accessory Worlds Coating Department: a. Compute how many cut mat sets were started in the...
-
28. Below is the model of a three-dimensional structure. Draw the front view of this model. Each cube is exactly inch in length, width, and height. Use a scale of 2:1. (2 marks) Back Top Front Left...
-
A national catalog and Internet retailer has three warehouses and three major distribution centers located around the country. Normally, items are shipped directly from the warehouses to the...
-
If P ( A ) = 0.30, P ( B ) = 0.40 and P ( A and B ) = 0.20, then P ( A / B ) is: A. 0.67 B. 0.12 C. 0.50 D. 0.08
-
CountVowels specs ** CountVowels Ask the user to enter a string. Print how many different vowels there are in the string (e.g. "aaaee" has 2 different vowels). Ignore case, i.e. 'A' and 'a' are...
-
Suppose the f(x, y) is a smooth function and that its partial derivatives have the values, fx (8,-7) = 3 and fy(8,-7)=5. Given that f(8, -7) = 0, use this information to estimate the value of...
-
#1: Moonshine Inc. has a positive retention ratio. If sales grew by 10 percent, the firm would have a negative projected EFN. a) What does this tell you about the firm's Internal Growth Rate (IGR)?...
-
What privacy or publicity concerns might a company have about the discovery process in traditional litigation?
-
You place $100 into a fund at the end of each month for 3 years. The annual nominal interest rate is 2% in the first year, 2.5% in the second year, and 3% in the third year. (a) Using a spreadsheet...
-
A company 'A' owns 49.9% of company 'S'. Another company 'B' owns 51.1% of S. B sells 'S' to another company for a gain with a higher FMV than book value and recognizes goodwill. Does 'A' have to...
-
Establish identity. cos( + k) = (-1)k cos , k any integer
-
The general ledger of Hensley Company contained the following Accounts Payable control account (in T-account form). Also shown is the related subsidiary ledger. Instructions (a) Indicate the missing...
-
On January 1, 2017, the ledger of Accardo Company contains the following liability accounts. Accounts Payable ....... $52,000 Sales Taxes Payable....... 7,700 Unearned Service Revenue..... 16,000...
-
Ling Corporation uses the equity method to account for its ownership of 35% of the common stock of Gorman Packing. During 2017, Gorman reported a net income of $80,000 and declares and pays cash...
-
On 1 January 2022, Markon plc took out a finance lease for new plant. Details of the lease are as follows: The business has a reporting-year-end date of 31 December. The non-cancellable part of the...
-
The financial statements for Harridges Ltd are given below for the two years ended 30 June 2020 and 2021. Harridges Limited operates a department store in the centre of a small town. Dividends were...
-
Identify and discuss three ratios that are likely to be affected by a business overtrading.

Study smarter with the SolutionInn App


