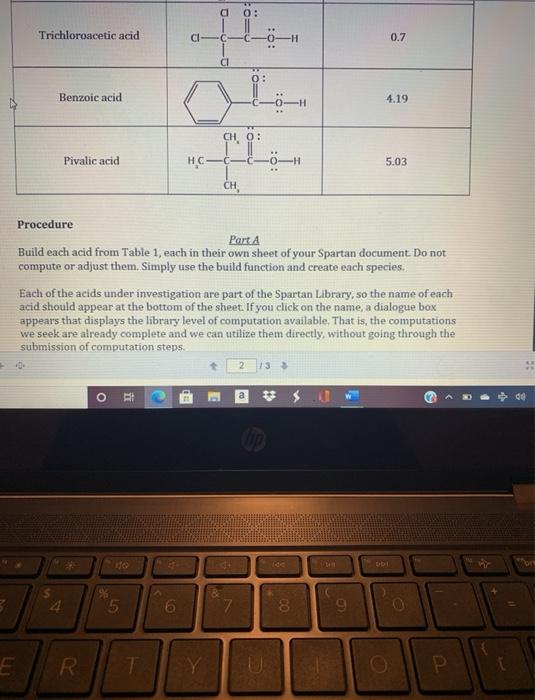
Table 1. Acidities and structure of the carboxylic acids of interest. Note that each acid contains...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
Table 1. Acidities and structure of the carboxylic acids of interest. Note that each acid contains the same protic bond....an 0-H bond. Acid Formic acid Acetic acid Chloroacetic acid Dichloroacetic acid Structure TV 0: H-C101H ww 0: H- H H 0: [[]][C] [COH C 0: Įdo T pka 3.75 4.75 2.85 1.48 Trichloroacetic acid Benzoic acid Procedure +0. E Pivalic acid 4 * R O ii do 5 D Part A Build each acid from Table 1, each in their own sheet of your Spartan document. Do not compute or adjust them. Simply use the build function and create each species. T Cl- Each of the acids under investigation are part of the Spartan Library, so the name of each acid should appear at the bottom of the sheet. If you click on the name, a dialogue box appears that displays the library level of computation available. That is, the computations we seek are already complete and we can utilize them directly, without going through the submission of computation steps. 6 HC- Cl Y CH 0: CH, 0: 7 2 -H 13 # $.0 140 8 1 9 0.7 4.19 5.03 bb1 O 0 S P br For your conclusion to the above exploration, discuss the trends you see in acid strength based upon the structure of the acid. Table 1. Acidities and structure of the carboxylic acids of interest. Note that each acid contains the same protic bond....an 0-H bond. Acid Formic acid Acetic acid Chloroacetic acid Dichloroacetic acid Structure TV 0: H-C101H ww 0: H- H H 0: [[]][C] [COH C 0: Įdo T pka 3.75 4.75 2.85 1.48 Trichloroacetic acid Benzoic acid Procedure +0. E Pivalic acid 4 * R O ii do 5 D Part A Build each acid from Table 1, each in their own sheet of your Spartan document. Do not compute or adjust them. Simply use the build function and create each species. T Cl- Each of the acids under investigation are part of the Spartan Library, so the name of each acid should appear at the bottom of the sheet. If you click on the name, a dialogue box appears that displays the library level of computation available. That is, the computations we seek are already complete and we can utilize them directly, without going through the submission of computation steps. 6 HC- Cl Y CH 0: CH, 0: 7 2 -H 13 # $.0 140 8 1 9 0.7 4.19 5.03 bb1 O 0 S P br For your conclusion to the above exploration, discuss the trends you see in acid strength based upon the structure of the acid.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
(a) Which of the carboxylic acids in Practice Problem 17.6 could be prepared by a nitrile synthesis as well? In problem 17.6 (b) Which synthesis, Grignard or nitrile, would you choose to prepare OH ...
-
The function and structure of the digestive tracts of the cow, horse, and human fit the diets of these animals. The grazers eat primarily leafy vegetation. Omnivores, however, consume more calories...
-
Describe the location and structure of the pituitary gland.
-
Consider the example in Exhibit 5.5. Can you think of anything else you might do with that example that would be helpful to the ultimate decisionmaker? exhibit 5.5 Decision Tree Analysis Using Net...
-
On May 1, 2012, Bovar Company began the manufacture of a new paging machine known as Dandy. The company installed a standard costing system to account for manufacturing costs. The standard costs for...
-
Q2. An engine is operating on the Atkinson cycle. The volumetric compression ratio is r = 9.2 and the pressure ratio is rp = 2.1. Take k = 1.4, cp = 1.005kJ/kgK. a) Draw the air-standard Atkinson...
-
You are considering three alternative investments in bonds. The bonds have different times to maturity, but carry the same default risk. You would like to gain an impression of the extent of price...
-
The following equation is sometimes used to forecast financial requirements: AFN = (A0*/S0) (S) (L0*/S0) (S) MS1 (1 POR) What key assumption do we make when using this equation? Under what...
-
This class is Management Information Systems which is also Management. The class is Management I don't know what else you need?
-
6. You have implemented a butterfly spread whereby the strategy will make money if the stock ends up between $44 and $56 (8 months from now). The stock currently trades at $48, its historical...
-
In 2 0 0 7 , Slater & Gordon ( S&G ) became the world s first law firm listed on a stock exchange. S&G is headquartered in Melbourne, where William Slater and Hugh Gordon founded it in 1 9 3 5 ....
-
I Question 1 (60 Points) Find the intrinsic value of a share of stock XYZ using the two-stage dividend discount model. The data for the valuation model has been obtained from the Value Line Research...
-
The rational expression equivalent to 2x+3 x +4 8 + is
-
Why are the variables of diversity and equity important to consider when developing a comprehensive discipline management plan that aligns to the school's vision and enhances the school's culture?...
-
Elizabeth's regular hourly wage rate is $22, and she receives an hourly rate of $33 for work in excess of 40 hours. During a January pay period, Elizabeth works 49 hours. Elizabeth's federal income...
-
From these websites: https://writingcommons.org/section/mindset/ https://www.psychologytoday.com/us/blog/click-here-happiness/201904/15-ways-build-growth-mindset What strikes you as most valuable in...
-
Water, assumed to be a perfect liquid, has a density of 1000 kg/m and a specific heat of 4184 J/kgK. The water undergoes an adiabatic, frictionless process in which its pressure is raised from 103...
-
Digital Fruit is financed solely by common stock and has outstanding 25 million shares with a market price of $10 a share. It now announces that it intends to issue $160 million of debt and to use...
-
What is the solution of the time-dependent Schrdinger equation (x,t) for the total energy eigenfunction (x) = 2/a sin (3x/a) for an electron in a one-dimensional box of length 1.00 10 10 m? Write...
-
Evaluate the commentator [P x + P 2 x , P 2 x ] by applying the operators to an arbitrary function f (x).
-
The heat of fusion of water is 6.008 10 3 J mol 1 at its normal melting point of 273.15 K. Calculate the freezing point depression constant K f .
-
What are two inherent limitations of financial statement data?
-
Quick Ratio Calculate the company's quick ratio for 2019 and compare the result to the industry average. Use the following financial data for Brenner Instruments Sales revenue.. Cost of goods sold...
-
Accounts Receivable Turnover Calculate the company's accounts receivable turnover for 2019 and compare the result to the industry average. Use the following financial data for Brenner Instruments...

Study smarter with the SolutionInn App


