The figure provides the T-s diagram of a Carnot heat pump cycle for which the substance...
Fantastic news! We've Found the answer you've been seeking!
Question:

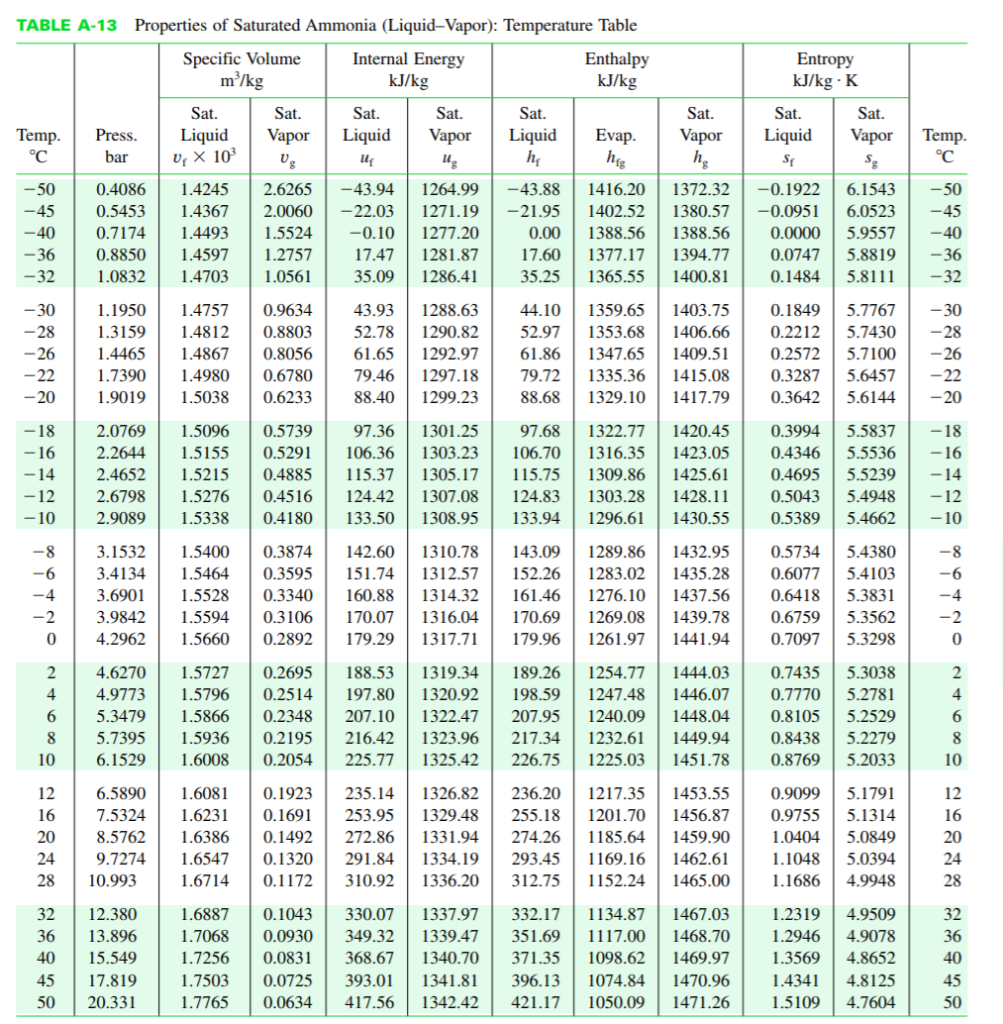
Transcribed Image Text:
The figure provides the T-s diagram of a Carnot heat pump cycle for which the substance is ammonia. Determine the net work input required, in kJ, for 50 cycles of operation and 0.1 kg of NH3 and whether any entropy is generated by the device. Given: x3 = 0.9. You may use either the tables posted under Handouts on the Google share (NH3.pdf) or EES to get the necessary ammonia properties. ΤΑ 2 20 bar 1 bar S TABLE A-13 Properties of Saturated Ammonia (Liquid-Vapor): Temperature Table Specific Volume Internal Energy m³/kg kJ/kg Temp. °℃ -50 -45 -40 -36 -32 -30 -28 -26 -22 -20 -8 -6 -4 -2 0 +680 DEGRE 5 -18 2.0769 -16 2.2644 1.5155 -14 -12 2.4652 1.5215 2.6798 1.5276 2.9089 1.5338 -10 10 Sat. Press. Liquid bar U X 10³ 12 16 2 4.6270 1.5727 0.2695 4 4.9773 1.5796 0.2514 0.2348 0.2195 0.2054 5.3479 1.5866 5.7395 1.5936 6.1529 1.6008 20 Sat. Vapor Ug 1.1950 1.4757 0.9634 1.3159 1.4812 0.8803 1.4465 1.4867 0.8056 1.7390 1.4980 0.6780 1.9019 1.5038 0.6233 Sat. Liquid U₂ Sat. Vapor Ug 32 12.380 36 13.896 40 15.549 45 17.819 50 20.331 1.5096 0.5739 97.36 1301.25 0.5291 106.36 1303.23 0.4885 115.37 1305.17 0.4516 124.42 1307.08 0.4180 133.50 1308.95 0.4086 1.4245 2.6265 -43.94 1264.99 -43.88 1416.20 0.5453 1.4367 2.0060 -22.03 1271.19 -21.95 1402.52 0.7174 1.4493 1.5524 -0.10 1277.20 0.00 1388.56 1388.56 0.8850 1.4597 1.2757 17.47 1281.87 17.60 1377.17 1394.77 1.0832 1.4703 1.0561 35.09 1286.41 35.25 1365.55 1400.81 44.10 1359.65 1403.75 52.97 1353.68 1406.66 61.86 1347.65 1409.51 79.72 1335.36 1415.08 88.68 1329.10 1417.79 43.93 1288.63 52.78 1290.82 61.65 1292.97 79.46 1297.18 88.40 1299.23 Sat. Liquid h₂ Enthalpy kJ/kg Evap. hig Sat. Vapor hg 1.6887 0.1043 330.07 1337.97 1.7068 0.0930 349.32 1339.47 1.7256 0.0831 368.67 1340.70 1.7503 0.0725 393.01 1341.81 1.7765 0.0634 417.56 1342.42 143.09 1289.86 1432.95 1283.02 1435.28 3.1532 1.5400 0.3874 142.60 1310.78 3.4134 1.5464 0.3595 151.74 1312.57 152.26 3.6901 1.5528 0.3340 160.88 1314.32 161.46 1276.10 1437.56 3.9842 1.5594 0.3106 170.07 1316.04 170.69 1269.08 1439.78 4.2962 1.5660 0.2892 179.29 1317.71 179.96 1261.97 1441.94 6.5890 1.6081 236.20 1217.35 1453.55 0.1923 235.14 1326.82 0.1691 253.95 1329.48 255.18 1201.70 1456.87 7.5324 1.6231 8.5762 1.6386 0.1492 272.86 1331.94 24 1.6547 0.1320 9.7274 28 10.993 291.84 1334.19 310.92 1336.20 274.26 1185.64 1459.90 293.45 1169.16 1462.61 312.75 1152.24 1465.00 1.6714 0.1172 97.68 1322.77 1420.45 106.70 1316.35 1423.05 115.75 1309.86 1425.61 124.83 1303.28 1428.11 133.94 1296.61 1430.55 1372.32 -0.1922 6.1543 1380.57 -0.0951 6.0523 0.0000 5.9557 0.0747 5.8819 0.1484 5.8111 188.53 1319.34 189.26 1254.77 1444.03 197.80 1320.92 198.59 1247.48 1446.07 207.10 1322.47 207.95 1240.09 1448.04 216.42 1323.96 217.34 1232.61 1449.94 225.77 1325.42 226.75 1225.03 1451.78 332.17 1134.87 1467.03 351.69 1117.00 1468.70 371.35 1098.62 1469.97 Entropy kJ/kg - K 396.13 1074.84 1470.96 421.17 1050.09 1471.26 Sat. Liquid SE Sat. Vapor Sg 0.1849 5.7767 0.2212 5.7430 0.2572 5.7100 0.3287 5.6457 0.3642 5.6144 0.3994 5.5837 0.4346 5.5536 0.4695 5.5239 0.5043 0.5389 0.5734 5.4380 0.6077 5.4103 0.6418 5.3831 0.6759 5.3562 0.7097 5.3298 0.7435 5.3038 0.7770 5.2781 0.8105 5.2529 0.8438 5.2279 0.8769 5.2033 0.9099 5.1791 0.9755 5.1314 1.0404 5.0849 1.1048 5.0394 1.1686 4.9948 1.2319 1.2946 1.3569 4.8652 1.4341 4.8125 1.5109 4.7604 Temp. °C 4.9509 4.9078 -50 -45 -14 -12 5.4948 5.4662 -10 -40 -36 -32 -30 -28 -26 -22 -20 -18 -16 -8 24823 55 5AN ONA ∞ -6 -4 -2 28 45 50 The figure provides the T-s diagram of a Carnot heat pump cycle for which the substance is ammonia. Determine the net work input required, in kJ, for 50 cycles of operation and 0.1 kg of NH3 and whether any entropy is generated by the device. Given: x3 = 0.9. You may use either the tables posted under Handouts on the Google share (NH3.pdf) or EES to get the necessary ammonia properties. ΤΑ 2 20 bar 1 bar S TABLE A-13 Properties of Saturated Ammonia (Liquid-Vapor): Temperature Table Specific Volume Internal Energy m³/kg kJ/kg Temp. °℃ -50 -45 -40 -36 -32 -30 -28 -26 -22 -20 -8 -6 -4 -2 0 +680 DEGRE 5 -18 2.0769 -16 2.2644 1.5155 -14 -12 2.4652 1.5215 2.6798 1.5276 2.9089 1.5338 -10 10 Sat. Press. Liquid bar U X 10³ 12 16 2 4.6270 1.5727 0.2695 4 4.9773 1.5796 0.2514 0.2348 0.2195 0.2054 5.3479 1.5866 5.7395 1.5936 6.1529 1.6008 20 Sat. Vapor Ug 1.1950 1.4757 0.9634 1.3159 1.4812 0.8803 1.4465 1.4867 0.8056 1.7390 1.4980 0.6780 1.9019 1.5038 0.6233 Sat. Liquid U₂ Sat. Vapor Ug 32 12.380 36 13.896 40 15.549 45 17.819 50 20.331 1.5096 0.5739 97.36 1301.25 0.5291 106.36 1303.23 0.4885 115.37 1305.17 0.4516 124.42 1307.08 0.4180 133.50 1308.95 0.4086 1.4245 2.6265 -43.94 1264.99 -43.88 1416.20 0.5453 1.4367 2.0060 -22.03 1271.19 -21.95 1402.52 0.7174 1.4493 1.5524 -0.10 1277.20 0.00 1388.56 1388.56 0.8850 1.4597 1.2757 17.47 1281.87 17.60 1377.17 1394.77 1.0832 1.4703 1.0561 35.09 1286.41 35.25 1365.55 1400.81 44.10 1359.65 1403.75 52.97 1353.68 1406.66 61.86 1347.65 1409.51 79.72 1335.36 1415.08 88.68 1329.10 1417.79 43.93 1288.63 52.78 1290.82 61.65 1292.97 79.46 1297.18 88.40 1299.23 Sat. Liquid h₂ Enthalpy kJ/kg Evap. hig Sat. Vapor hg 1.6887 0.1043 330.07 1337.97 1.7068 0.0930 349.32 1339.47 1.7256 0.0831 368.67 1340.70 1.7503 0.0725 393.01 1341.81 1.7765 0.0634 417.56 1342.42 143.09 1289.86 1432.95 1283.02 1435.28 3.1532 1.5400 0.3874 142.60 1310.78 3.4134 1.5464 0.3595 151.74 1312.57 152.26 3.6901 1.5528 0.3340 160.88 1314.32 161.46 1276.10 1437.56 3.9842 1.5594 0.3106 170.07 1316.04 170.69 1269.08 1439.78 4.2962 1.5660 0.2892 179.29 1317.71 179.96 1261.97 1441.94 6.5890 1.6081 236.20 1217.35 1453.55 0.1923 235.14 1326.82 0.1691 253.95 1329.48 255.18 1201.70 1456.87 7.5324 1.6231 8.5762 1.6386 0.1492 272.86 1331.94 24 1.6547 0.1320 9.7274 28 10.993 291.84 1334.19 310.92 1336.20 274.26 1185.64 1459.90 293.45 1169.16 1462.61 312.75 1152.24 1465.00 1.6714 0.1172 97.68 1322.77 1420.45 106.70 1316.35 1423.05 115.75 1309.86 1425.61 124.83 1303.28 1428.11 133.94 1296.61 1430.55 1372.32 -0.1922 6.1543 1380.57 -0.0951 6.0523 0.0000 5.9557 0.0747 5.8819 0.1484 5.8111 188.53 1319.34 189.26 1254.77 1444.03 197.80 1320.92 198.59 1247.48 1446.07 207.10 1322.47 207.95 1240.09 1448.04 216.42 1323.96 217.34 1232.61 1449.94 225.77 1325.42 226.75 1225.03 1451.78 332.17 1134.87 1467.03 351.69 1117.00 1468.70 371.35 1098.62 1469.97 Entropy kJ/kg - K 396.13 1074.84 1470.96 421.17 1050.09 1471.26 Sat. Liquid SE Sat. Vapor Sg 0.1849 5.7767 0.2212 5.7430 0.2572 5.7100 0.3287 5.6457 0.3642 5.6144 0.3994 5.5837 0.4346 5.5536 0.4695 5.5239 0.5043 0.5389 0.5734 5.4380 0.6077 5.4103 0.6418 5.3831 0.6759 5.3562 0.7097 5.3298 0.7435 5.3038 0.7770 5.2781 0.8105 5.2529 0.8438 5.2279 0.8769 5.2033 0.9099 5.1791 0.9755 5.1314 1.0404 5.0849 1.1048 5.0394 1.1686 4.9948 1.2319 1.2946 1.3569 4.8652 1.4341 4.8125 1.5109 4.7604 Temp. °C 4.9509 4.9078 -50 -45 -14 -12 5.4948 5.4662 -10 -40 -36 -32 -30 -28 -26 -22 -20 -18 -16 -8 24823 55 5AN ONA ∞ -6 -4 -2 28 45 50
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these mechanical engineering questions
-
A mixing chamber receives 5 kg/min ammonia as saturated liquid at 20C from one line and ammonia at 40C, 250 kPa from another line through a valve. The chamber also receives...
-
A mixing chamber receives 10 lbm/min ammonia as saturated liquid at 0 F from one line and ammonia at 100 F, 40 lbf/in 2 from another line through a valve. The chamber also receives 340 Btu/min energy...
-
A mixing chamber receives 5 kg/min ammonia as saturated liquid at 20C from one line and ammonia at 40C, 250 kPa from another line through a valve. The chamber also receives 325 kJ/min energy as heat...
-
In Exercises find the indefinite integral. sech(2x1) dx
-
Duncan Motors is introducing a new product that it expects will increase its net operating income by $300,000. Duncan Motors has a 34 percent marginal tax rate. This project will also produce $50,000...
-
Reference frame S' passes reference frame S with a certain velocity as in Figure. Events 1 and 2 are to have a certain spatial separation x' according to the S' observer. However, their temporal...
-
Identify the research scenario, including the general area of focus. Develop a hypothetical research scenario that would necessitate the use of the Mixed Method A-B-A Design. The research will be...
-
Chandler Ltd. estimates sales for the second quarter of 2014 will be as follows. Month Units April ...... 2,550 May ....... 2,475 June ....... 2,390 The target ending inventory of finished products...
-
A quadratic Bezier curve is defined by 3 points (PO, P1 and P2 The curve starts at PO and stops at P2. Other points are defined as P(t) in terms of t, PO, P1 and P2. Find point positions in curve for...
-
1. As much as possible, assess the extent to which PetroChinas accounting principles conform to IASB standards. 2. How reliable is your assessment? 3. What further information would help your...
-
Ex. (12): Use Divergence theorem to evaluate ff, F. ds, where F = x i+yj+z k and S is the surface of the sphere x + y + z = a?
-
How do you do deductions for city income taxes at a 1.0% from the employee grss wage?
-
Why should auditors expect to be more challenged on their findings?
-
How to recognize communication quality (accurate, objective, clear, concise, constructive, complete, and timely) and elements (objectives, scope, conclusions, recommendations, and action plan)...
-
How to calculate % unit increased decreased by sell thru report?
-
The current stock price of Well-Tempered Flugelhorns (WTF) is $40 with an instantaneous standard deviation of 20%. If the risk-free rate is 3%, what is the value of a put option with an exercise...
-
The U.S. economy has started to face a slowdown this year (2023). Among other factors, investment and consumption expenditures have decreased with less credit available (Hint: negative demand shock)...
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
List the utilization of geothermal energy. Which use is most common?
-
Saturated refrigerant-134a vapor at 15 psia is compressed reversibly in an adiabatic compressor to 80 psia. Determine the work input to the compressor. What would your answer be if the refrigerant...
-
The thrust developed by the engine of a Boeing 777 is about 380 kN. Assuming choked flow in the nozzles, determine the mass flow rate of air through the nozzle. Take the ambient conditions to be 220...
-
To test \(H_{0}: \sigma=50\) versus \(H_{1}: \sigma <50\), a random sample of size \(n=24\) is obtained from a population that is known to be normally distributed. (a) If the sample standard...
-
In Example 2 from Section 10.3, the quality-control engineer for M\&MMars tested whether the mean weight of fun-size Snickers was 20.1 grams. Suppose that the standard deviation of the weight of the...
-
(a) Determine the critical values for a right-tailed test of a standard population deviation with 18 degrees of freedom at \(\alpha=0.1\) level of significance. (b) Determine the critical values for...

Study smarter with the SolutionInn App


