Take one of the following sets X and Y of chemicals? X ? Y (i) Copper sulphate
Fantastic news! We've Found the answer you've been seeking!
Question:
- Take one of the following sets X and Y of chemicals?X ? Y(i) Copper sulphate ? Sodium carbonate(ii) Barium chloride ? Sodium sulphate(iii) Lead nitrate ? Sodium chloride
- Prepare separately 95% solution of any one pair of substances listed under X and Y in water.
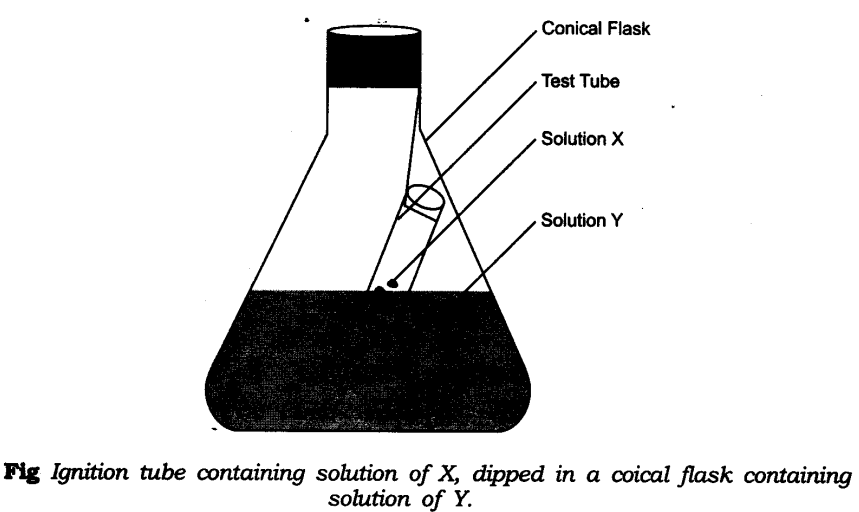
- Take little amount of solution of Y in a conical flask and some solution of X in an ignition tube.
- Hang the ignition tube in the flask carefully; see that the solutions do not get mixed.
- Put a cork on the flask as shown in figure.
- Weigh the flask with its contents carefully.
- Now tilt and swirl the flask, so that the solutions X and Y get mixed.
- Weigh again.
- What happens in the reaction flask?
- Do you think that a chemical reaction has taken place?
- Why should we put a cork on the mouth of the flask?
- Does the mass of the flask and its contents change?
Related Book For 

Posted Date:




