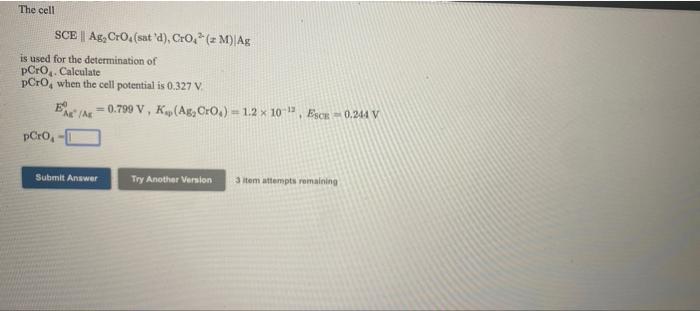
The cell SCE | Ag, Cro,(sat 'd), Cro,* (z M)|Ag is used for the determination of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The cell SCE | Ag, Cro,(sat 'd), Cro,* (z M)|Ag is used for the determination of pCro,. Calculate pCro, when the cell potential is 0.327 V. E e /Ar- 0.799 V, K(Ag, Cro,) = 1.2 x 10 , EsCR 0.244 V pCro, Submit Answer Try Another Version 3 tem attempts remaining The cell SCE | Ag, Cro,(sat 'd), Cro,* (z M)|Ag is used for the determination of pCro,. Calculate pCro, when the cell potential is 0.327 V. E e /Ar- 0.799 V, K(Ag, Cro,) = 1.2 x 10 , EsCR 0.244 V pCro, Submit Answer Try Another Version 3 tem attempts remaining
Expert Answer:
Answer rating: 100% (QA)
ans Representation ofcen 2 suturrate d acam Ing lest Side part The ... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
The cell SCE((Ag2CrO4 (sat'd), (x M) (Ag is used for the determination of pCrO4. Calculate pCrO4 when the cell potential is 0.389 V.
-
Calculate Z and V for ethylene at 25(C and 12 bar by the following equations: (a) The truncated virial equation [Eq. (3.401] with the following experimental values of virial coefficients: (b) The...
-
Calculate Z and V for ethane at 50(C and 15 bar by the following equations: (a) The truncated virial equation [Eq. (3.401] with the following experimental values of virial coefficients: (b) The...
-
Show the income statement, cash flow statement and balance sheet of the company at 31/12/2020 if: 1. The company start its operations on June 1st, 2020. Over the period, it sells 50 laptop for $400....
-
What are the major organization-focused, group-focused, and individual-focused OD intervention methods?
-
Why is it important for managers and all employees to interact with people who are different from themselves?
-
On 1 July 2025, McGregor and Roberts decided to amalgamate their businesses and to share profits equally. Financial information at that date was as follows. At 1 July 2025, McGregors accounts...
-
You are performing an experiment to measure the specific internal energy of a gas relative to a reference state of 25?C and 1 atm (at which conditions U is arbitrarily set equal to 0). The gas is...
-
4. 5. A heavy particle is projected with a velocity at an angle with the horizontal into the uniform gravitational field. The slope of the trajectory of the particle varies with its horizontal...
-
Employees sometimes make mistakes and errors in recording accounting transactions. Is this considered fraud?
-
Which method allocates joint costs on the basis of each product's relative sales value at the split-off point: a The estimated net realizable value method b the sales value at the split-off method c...
-
You are using a steel crowbar to lift a big rock, with a smaller stone as a fulcrum at the center of the crowbar, but you are not quite able to lift the rock. What can you change about this setup to...
-
SAT scores are normally distributed with a mean of 1518 and a standard deviation of 325. Decide whether the statement makes sense (or is clearly true) or does not make sense (or is clearly false)....
-
A certain car engine delivers enough force to create \(380 \mathrm{~N} \cdot \mathrm{m}\) of torque when the engine is operating at 3200 revolutions per minute. Calculate the average power delivered...
-
Helicopters have a small tail rotor as well as the large main rotor. Why is the tail rotor needed?
-
The specifications for tightening the bolts on a car engine's valve cover are given in terms of torque rather than in terms of how much force should be exerted on the tightening wrench. Why is torque...
-
is a traditional function of the MIS department. O Providing technical services O Educating the MIS staff about the business O Initiating and designing specific strategic IS Proactively using...
-
The text defined intrinsic value as the value of an asset given a hypothetically complete understanding of the assets investment characteristics. Discuss why hypothetically is included in the...
-
The following are summaries of the cash book and bank accounts of J. Duncan who does not keep his books using the double entry system. You are required to: (a) Calculate the value of J. Duncan's...
-
Read the following and answer the questions below. On 31 December 2015 the bank column of C. Tench's cash book showed a debit balance of 1,500. The monthly bank statement written up to 31 December...
-
The bank columns in the cash book for June 2016 and the bank statement for that month for D. Hogan are as follows: You are required to: () Write the cash book up to date to take the above into...

Study smarter with the SolutionInn App

