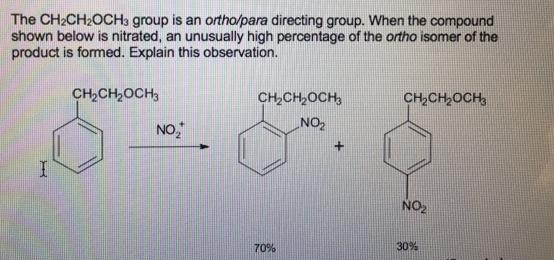
The CH2CH2OCH3 group is an ortho/para directing group. When the compound shown below is nitrated, an...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The CH2CH2OCH3 group is an ortho/para directing group. When the compound shown below is nitrated, an unusually high percentage of the ortho isomer of the product is formed. Explain this observation. CH2CH2OCH3 CH,CH,OCH, CH,CH,OCH NO, ZON NO2 70% 30% The CH2CH2OCH3 group is an ortho/para directing group. When the compound shown below is nitrated, an unusually high percentage of the ortho isomer of the product is formed. Explain this observation. CH2CH2OCH3 CH,CH,OCH, CH,CH,OCH NO, ZON NO2 70% 30%
Expert Answer:
Answer rating: 100% (QA)
1 2 In second question looking at the relative rate for the ortho position in the given ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
When the following compound is heated, a product is formed that shows an infrared absorption band at 1715 cm-1. Draw the structure of the product. CH3 CH
-
Compound B, an isomer of A (Problem 20.47), is also soluble in dilute HCl. The IR spectrum of B shows no bands in the 3300-3500-cm-1 region. The broadband proton-decoupled 13C spectrum of B is given...
-
An ester is a compound formed by a condensation reaction between a carboxylic acid and an alcohol. Read the discussion of esters in Section 24.4 and then give an example of a reaction forming an...
-
An SAT prep course claims to increase student scores by more than 60 points, on average. To test this claim, 9 students who have previously taken the SAT are randomly chosen to take the prep course....
-
A stock has an exercise (strike) price of $40. a. If the stock price goes to $41.50, is the exchange likely to add a new strike price? b. If the stock price goes to $42.75 is the exchange likely to...
-
What are the major advantages to a company of implementing an effective enterprise risk management process?
-
Assume HPC, Inc., began December with 60 units of inventory that cost a total of $720. During December, HPC, Inc., purchased and sold goods as follows: HPC, Inc., uses perpetual inventory. Under the...
-
From the following, calculate the dividends for common and preferred stock: 8% fully participating preferred stock. The board declared a $210,000 dividend. Preferred stock 5,000 shares, $50 par...
-
You have successfully negotiated the draft budget with each department head of the Hotel which has no been approved by the director and implemented 6 weeks ago. You have finalised the financial data...
-
Kathy wants to buy a condominium selling for $95,000. The taxes on the property are $1500 per year, and homeowners insurance is $336 per year. Kathys gross monthly income is $4000. She has 15 monthly...
-
Consider a satellite in stationary orbit that beams a signal earthward with a power of 4.9 kilowatts and a beamwidth covering a circular region with diameter 193 km on Earth. What is the root-mean-...
-
1. Suppose that under competitive conditions the market for talent can be represented by the following functions: Supply of Labor: P = 50 + 3Q Demand for Labor: P = 200 - 3Q Find and graph the...
-
Present and future values of $1 at 9% are presented below. PV of $1 FV of $1 1 0.91743 1.09000 PVA of $1 0.91743 FVAD of $1 FVA of $1 1.0900 1.0000 2 0.84168 1.18810 1.75911 2.2781 2.0900 3 0.77218...
-
There are a number of reasons why human resources managers might want to develop a strategy for providing employee voice. Here are some possibilities: Learning about employee ideas for improved...
-
Consider the following information relating to the nonmonetary exchange of assets. Fair value of assets given up $120,000 Fair value of assets acquired 150,000 Carrying value of assets given up...
-
The government of Example Land is considering the introduction of a series of labor market reforms that will restore employee and union rights to collective bargaining, introduce industry-level...
-
A = = -1 -2 -1 0 B = 1 0 1 - Ex: 42 AB =
-
Find the velocity, acceleration, and speed of a particle with the given position function. r(t) = (t 2 , sin t - t cos t, cos t + t sin t), t > 0
-
a. How many asymmetric carbons does cholesterol have? b. What is the maximum number of stereoisomers that cholesterol can have? c. How many of these stereoisomers are found in nature? CHj H3C CH3 HC...
-
Draw a reaction coordinate diagram for a reaction in which a. the product is thermodynamically unstable and kinetically unstable. b. the product is thermodynamically unstable and kinetically stable.
-
Explain why thymine cannot be deaminated.
-
The man has a mass of 78 kg and stands motionless at the end of the diving board. If the board has the cross section shown, determine the maximum normal strain developed in the board. The modulus of...
-
The steel rod having a diameter of 1 in. is subjected to an internal moment of M = 300lbft. Determine the stress created at points A and B. Also, sketch a three-dimensional view of the stress...
-
The steel beam has the cross-sectional area shown. If w=5 kip/ft, determine the absolute maximum bending stress in the beam. W 8 8 ft- 8 ft W ft- -8 ft- .8 in. 0.30 in. 0.3 in.- 10 in. 0.30 in.

Study smarter with the SolutionInn App


