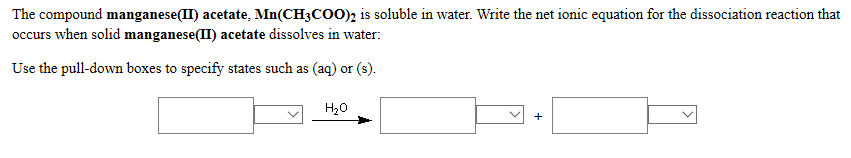
The compound manganese(II) acetate, Mn(CH3COO)2 is soluble in water. Write the net ionic equation for the...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
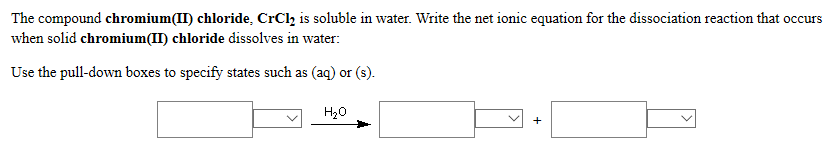
The compound manganese(II) acetate, Mn(CH3COO)2 is soluble in water. Write the net ionic equation for the dissociation reaction that occurs when solid manganese(II) acetate dissolves in water: Use the pull-down boxes to specify states such as (aq) or (s). H₂O + The compound sodium chloride, NaCl is soluble in water. Write the formulas for the ions that interact with water as shown in the models when solid sodium chloride dissolves in water: (Do not include the stoichiometries in the formulas.) Interaction Model Ion Formula The compound chromium(II) chloride, CrCh₂ is soluble in water. Write the net ionic equation for the dissociation reaction that occurs when solid chromium(II) chloride dissolves in water: Use the pull-down boxes to specify states such as (aq) or (s). H₂O + The compound manganese(II) acetate, Mn(CH3COO)2 is soluble in water. Write the net ionic equation for the dissociation reaction that occurs when solid manganese(II) acetate dissolves in water: Use the pull-down boxes to specify states such as (aq) or (s). H₂O + The compound sodium chloride, NaCl is soluble in water. Write the formulas for the ions that interact with water as shown in the models when solid sodium chloride dissolves in water: (Do not include the stoichiometries in the formulas.) Interaction Model Ion Formula The compound chromium(II) chloride, CrCh₂ is soluble in water. Write the net ionic equation for the dissociation reaction that occurs when solid chromium(II) chloride dissolves in water: Use the pull-down boxes to specify states such as (aq) or (s). H₂O +
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Write the net ionic equation for the reaction of BaCl2(aq) and Na2SO4(aq). You may have to consult the solubility rules.
-
Write the net ionic equation for the reaction of KCl(aq) and NaC2H3O2(aq). You may have to consult the solubility rules.
-
Write the net ionic equation for the reaction of Fe2(SO4)3(aq) and Sr(NO3)2(aq). You may have to consult the solubility rules.
-
For the given values, determine the quadrant(s) in which the terminal side of the angle lies. cos = 0.5000
-
What potential security problems do you see in the increasing use of intranets and extranets in business? What might be done to solve such problems? Give several examples.
-
Burrito Bell issued a series of $1,000 bonds eight years ago with an annual coupon rate of $100. The bonds mature 12 years from now. If an investor requires a 6 percent return on this investment,...
-
Find the critical value f0.05 for F7,20.
-
Marble Construction estimates that its WACC is 10% if equity comes from retained earnings. However, if the company issues new stock to raise new equity, it estimates that its WACC will rise to 10.8%....
-
Balraj recently opened an electronic repair store and in the first 3 months has had great success. In fact he just closed the deal on a contract that will have him servicing the town library's...
-
Chickadee Ski Club (CSC) is located in southern Ontario near large urban centres but not far enough north to guarantee a full winter of snow on the ground. The club's revenue and profit jump in years...
-
HIRARC is the fundamental key element in managing occupational risk. This activity must be supported with other elements to ensure the effectiveness of risk control, making the workplace safer and...
-
Make a Biblical Appraisal of Christian Counselling and discuss Discuss the goals of Christian counselling - what makes Christian counselling unique?
-
If Baxter Inc. has annual sales (all credit sales) of $5,000,000, and receivables equal to 10% of sales, how many days of sales outstanding are there ?
-
A year ago you purchased a 10-yr 0 coupon bond with a YTM of 5% and FV of $1K. If you sell the bond, which now has YTM of 8% then what is the one yr holding period return on investment?
-
Provide specific data on Spotify's revenue over the past five years. Highlight any significant growth or fluctuations. Compare the revenue growth of Spotify with its key competitors, such as Apple...
-
For the current fiscal year, Purchases were $187,000, Purchase Returns and Allowances were $4,200 and Freight In was $10,500. If the beginning merchandise inventory was $98,000 and the ending...
-
With a short response to the following discussion posts Post #1 As a project manager, the entire project managment process was new to me. Learning how to create the project charter, about project...
-
Define cultural intelligence. Cite the books or journal articles you found in Capella's library. Explain why cultural intelligence is important for HR practitioners and other organizational managers.
-
Concentrated aqueous ammonia contains 1.00 mol NH3 dissolved in 2.44 mol H2O. What is the mole fraction of NH3 in concentrated aqueous ammonia? What is the molal concentration of NH3?
-
Two compounds have the formula Pt(NH3)2Cl2. (Compound B is cisplatin). They have square planar structures. One is expected to have a dipole moment; the other is not. Which one would have a dipole...
-
Any object, be it a space satellite or a molecule, must attain an initial upward velocity of at least 11.2 km/s in order to escape the gravitational attraction of the earth. What would be the kinetic...
-
Acorn Growth Limited is a small technology company listed on the TSX. To conserve cash, the company frequently settles obligations through the issuance of rights and options. Shares are now trading...
-
Convertible Debt, Three Cases: The following cases are independent: Case A On 1 November 20X1, Bertha Builders Limited issued a convertible bond that was convertible in 15 years' time into 78,000...
-
NewTech Ltd. has a 31 December fiscal year-end. The company issued convertible bonds on 1 July 20X4. The \(\$ 5,000,000\) bonds pay annual interest of \(8 \%\) each 30 June and mature on 30 June...

Study smarter with the SolutionInn App


