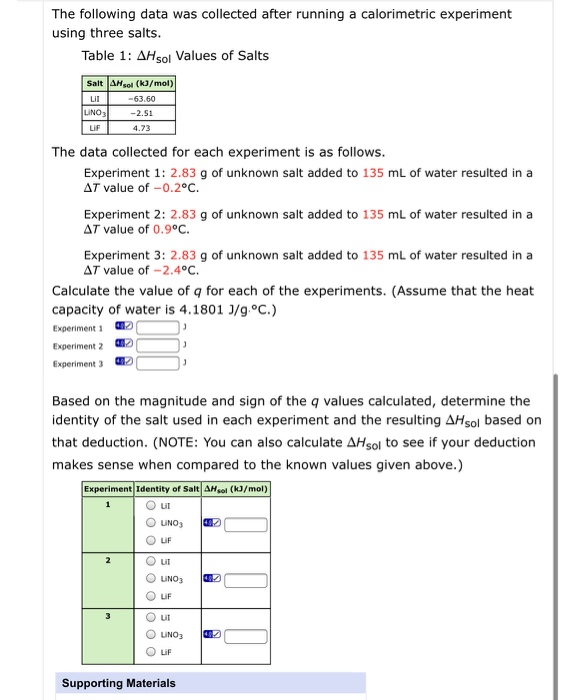
The following data was collected after running a calorimetric experiment using three salts. Table 1: AHsol...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The following data was collected after running a calorimetric experiment using three salts. Table 1: AHsol Values of Salts Salt AHsol (kJ/mol) Lil LINO3 -63.60 -2.51 LIF 4.73 The data collected for each experiment is as follows. Experiment 1: 2.83 g of unknown salt added to 135 ml of water resulted in a AT value of -0.2°C. Experiment 2: 2.83 g of unknown salt added to 135 mL of water resulted in a AT value of 0.9°c. Experiment 3: 2.83 g of unknown salt added to 135 mL of water resulted in a AT value of -2.4°c. Calculate the value of q for each of the experiments. (Assume that the heat capacity of water is 4.1801 J/g.°C.) Experiment 1 Experiment 2 Experiment 3 Based on the magnitude and sign of the q values calculated, determine the identity of the salt used in each experiment and the resulting AHsol based on that deduction. (NOTE: You can also calculate AHsol to see if your deduction makes sense when compared to the known values given above.) Experiment Identity of Salt AHsol (kɔ/mol) LINO3 LIF O ut O UNO3 LIF O ut O UNO3 Lif Supporting Materials O O 0 O O The following data was collected after running a calorimetric experiment using three salts. Table 1: AHsol Values of Salts Salt AHsol (kJ/mol) Lil LINO3 -63.60 -2.51 LIF 4.73 The data collected for each experiment is as follows. Experiment 1: 2.83 g of unknown salt added to 135 ml of water resulted in a AT value of -0.2°C. Experiment 2: 2.83 g of unknown salt added to 135 mL of water resulted in a AT value of 0.9°c. Experiment 3: 2.83 g of unknown salt added to 135 mL of water resulted in a AT value of -2.4°c. Calculate the value of q for each of the experiments. (Assume that the heat capacity of water is 4.1801 J/g.°C.) Experiment 1 Experiment 2 Experiment 3 Based on the magnitude and sign of the q values calculated, determine the identity of the salt used in each experiment and the resulting AHsol based on that deduction. (NOTE: You can also calculate AHsol to see if your deduction makes sense when compared to the known values given above.) Experiment Identity of Salt AHsol (kɔ/mol) LINO3 LIF O ut O UNO3 LIF O ut O UNO3 Lif Supporting Materials O O 0 O O
Expert Answer:
Answer rating: 100% (QA)
First of all Convert Ansol of each salt om KJmof to ... View the full answer

Related Book For 

Probability and Statistical Inference
ISBN: 978-0321923271
9th edition
Authors: Robert V. Hogg, Elliot Tanis, Dale Zimmerman
Posted Date:
Students also viewed these accounting questions
-
Although we often assume that the heat capacity of a substance is not temperature dependent, this is not strictly true, as shown by the following data for ice: Use these data to calculate graphically...
-
Among the data collected for the World Health Organization air quality monitoring project is a measure of suspended particles, in g/m3. Let X and Y equal the concentration of suspended particles in...
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
During the year, the total actual cost of inventory produced for The Wood Legacy was $1,500,000 (assume no beginning or ending inventory balances). Actual manufacturing overhead was $300,000. Direct...
-
Social Security payments to the elderly are adjusted every year in the following way: The government has in the past determined some average bundle of goods consumed by an average elderly person....
-
(1) True or false: (a) Angular velocity and linear velocity have the same dimensions. (b) All parts of a rotating wheel must have the same angular velocity. (c) All parts of a rotating wheel must...
-
For the ammonia product, carry out the development stage of the SGPDP as suggested in Section 27.3.
-
The commissioners of the Regents Park Commission Special Revenue Fund approved the following budget for calendar year 2012. Assume that the Unassigned fund balance at the beginning of the year was $...
-
If you want to invest in a portfolio that consists from two assets, stock X as 70% from this investment and stock Z as 30%. The expected rate of return on share X is 15% and for Z is 9%, the standard...
-
Design the 4-to-1 MUX two ways Write a Verilog module called mux4to1 to implement 4-to-1 multiplexer using functional descriptions and if-else blocks. Write another Verilog module called...
-
Water Play Pty Ltd sells inflatable water toys and other pool play items. You have been asked to prepare budgets for the quarter ended 31 March 2023 for the business based on the following...
-
Make a conclusion for this assignment if possibvle TIME CONTENT TEACHING/LEARNING ACTIVITIES RESOURCES/ MATERIALS 2mins Introduction We will begin by introducing ourselves and giving a brief overview...
-
Which of the following statements about licensing is true? a) When a product is licensed, the foreign partner will use the licensor's technology as a guide for home country business operations. b)...
-
On January 1, 2025, Wildhorse Corporation changed to the percentage-of-completion method of income recognition. This change is justified by Wildhorse Corporation. On December 31, 2024, Wildhorse's...
-
How do biogeographic barriers, such as mountain ranges, rivers, and oceans, influence patterns of species distribution and genetic divergence, and what insights can phylogeographic analyses offer...
-
Does the exponential expression represent growth or decay? 0.92(1.006)' The given exponential expression represents
-
What perspective on leadership focuses on the role communication plays in influencing and directing groups?
-
Write a paper about medication error system 2016.
-
In 2012, Red Rose tea randomly began placing 1 of 12 English porcelain miniature figurines in a l00-bag box of the tea, selecting from 12 nautical figurines. (a) On the average, how many boxes of tea...
-
Suppose that a third group of nurses was observed along with groups I and II of Exercise 9.2-1, resulting in the respective frequencies 130, 75, 136, 33, 61, and 65. Test H0: pi1 = pi2 = pi3, i = 1,...
-
Show that the expression in the exponent of Equation 4.5-2 is equal to the function q(x, y) given in the text.
-
In 2022, Mark purchased two separate activities. Information regarding these activities for 2022 and 2023 is as follows: The 2022 losses were suspended losses for that year. During 2023, Mark also...
-
In 2023, Julie, a single individual, reported the following items of income and deduction: Julie owns 100% and is an active participant in the rental real estate activity. What is her taxable income...
-
Jerry sprayed all of the landscaping around his business with a pesticide in June 2023. Shortly thereafter, all of the trees and shrubs unaccountably died. The FMV and the adjusted basis of the...

Study smarter with the SolutionInn App


