The dissolution of ammonium nitrate is given by the reaction: NH,NO,(s) NH (ag) + NO, (ag)...
Fantastic news! We've Found the answer you've been seeking!
Question:
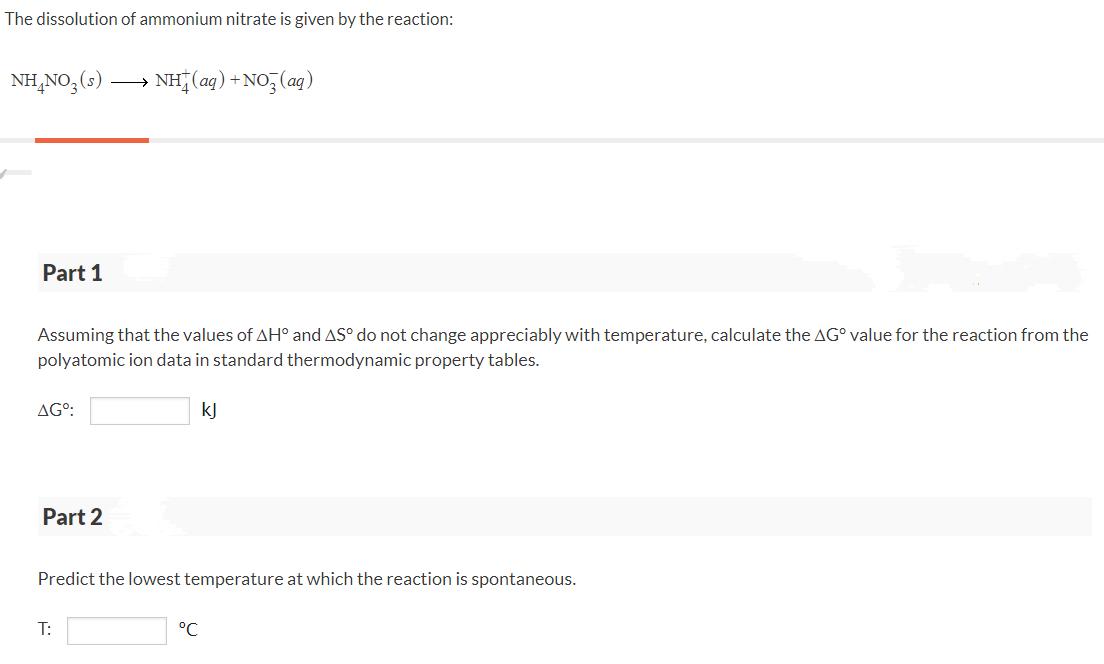
Transcribed Image Text:
The dissolution of ammonium nitrate is given by the reaction: NH,NO,(s) NH (ag) + NO, (ag) Part 1 Assuming that the values of AH° and AS° do not change appreciably with temperature, calculate the AG° value for the reaction from the polyatomic ion data in standard thermodynamic property tables. AG°: kJ Part 2 Predict the lowest temperature at which the reaction is spontaneous. T: °C The dissolution of ammonium nitrate is given by the reaction: NH,NO,(s) NH (ag) + NO, (ag) Part 1 Assuming that the values of AH° and AS° do not change appreciably with temperature, calculate the AG° value for the reaction from the polyatomic ion data in standard thermodynamic property tables. AG°: kJ Part 2 Predict the lowest temperature at which the reaction is spontaneous. T: °C
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Ammonium nitrate is an oxidizing agent and can give rise to explosive mixtures. A mixture of 2.00 mol of powdered aluminum and 3.00 mol of ammonium nitrate crystals reacts exothermically, yielding...
-
As mentioned in Chapter 3 (p. 108), ammonium nitrate (NH4 NO3) is the most important nitrogen-containing fertilizer in the world. Describe how you would prepare this compound, given only air and...
-
Predict a possible monatomic ion for Element 117, Uus. Given the spherical models below, which should be labeled atom and which should be labeled ion?
-
A billboard at the top of a building is being illuminated by a projection light (L) that is 103 feet from the base of the building as shown in the figure. Determine how tall the billboard if its...
-
Electronic transactions can result in the creation and combination of databases containing sensitive business and personal information. Those who provide this information are naturally concerned...
-
Exhibits 10-10A and B contain excerpts from the University of Torontos annual report for its 2012/13 fiscal year. EXHIBIT 10-10B EXCERPT FROM UNIVERSITY OF TORONTOS 2012/13 ANNUAL REPORT On December...
-
Defendant Kenneth Blake was married to Charlene Hinton-Blake, who died in 2012. Three of Hinton-Blakes sisters, including Yvonne Hinton, assisted her with day-to-day care during a four-year period of...
-
Speedy Oil provides a single-channel automobile oil change and lubrication service. Customers provide an arrival rate of 2.5 cars per hour. The service rate is 5 cars per hour. Assume that arrivals...
-
in the COVID pandemic of 2020, the world's largest pharmaceutical companies were quick to develop vaccines. Would a market composed of many small companies have been able to respond as fast?
-
The equation of motion for a pendulum whose base is accelerating horizontally with an acceleration a(t) is L + g sin = a(t)cos Suppose that g = 9.81 m/s2, L = 1 m, and (0) = 0. Solve for and plot ...
-
Let and beta be the roots of x-x-1= 0, with > beta. For all positive integers n, define n a" - B a-B an = correct? A. C. b n=110n " 8 89 n1, b = 1 and bn = an- 1 +an+1, n 2. Then which of the...
-
Describe a real-life personal finance scenario that encompasses the narrative story of what the personal financial activity was (getting a bank loan, buying a car, getting somebody to co-sign your...
-
14. Hartley Corporation is in possession of Weber Corporation's promissory note, which has a principal amount of $40,000, a term of 180 days, and carries an annual interest rate of 4%. The journal...
-
What is the purpose of Bridgewater's All-weather portfolio? Question 3 Answer a. To maximise the return of the portfolio b. To have a portfolio that can be invested under all weather conditions c. To...
-
Jay and Mary want to start their own olive oil company. Jay is a specialist in olive oil, but he lacks money. Despite her lack of experience in olive oil production, Mary is willing to provide all...
-
Write a 3-4 page review of the role that SOC reporting provides to those outsourcing an accounting function such as payroll. Demonstrate that you have reviewed the resources provided and have an...
-
When pricing an item such as an auto for sale, first an individual may consider how much has been invested in the vehicle. 1. Discuss the relationship or lack of relationship, been the amount...
-
Write the binomial probability in words. Then, use a continuity correction to convert the binomial probability to a normal distribution probability. P(x 110)
-
Using scientific notation, convert: a. 6.20 km to m b. 1.98 ns to s c. 2.54 cm to m d. 5.23 g to g
-
The main ingredient in many phosphate fertilizers is Ca(H2PO4)2 H2O. If a fertilizer is 17.1% P (by mass), and all of this phosphorus is present as Ca(H2PO4)2 H2O, what is the mass percentage of...
-
Boron trifluoride, BF3, reacts with ammonia, NH3, to form an addition compound, BF3NH3. Describe the geometries about the B and the F atoms in this compound. Describe the hybridization on these two...
-
The business staff of the law firm Frampton, Davis & Smythe has constructed the following report that breaks down the firms overall results for last month into two business segmentsfamily law and...
-
The Excel worksheet form that appears below is to be used to recreate portions of Review Problem 1 relating to Dexter Corporation. Download the workbook containing this form from Connect, where you...
-
Millard Corporation is a wholesale distributor of office products. It purchases office products from manufacturers and distributes them in the West, Central, and East regions. Each of these regions...

Study smarter with the SolutionInn App


