The energies of the 3 lowest states of an atom are E = -14 eV, E,...
Fantastic news! We've Found the answer you've been seeking!
Question:
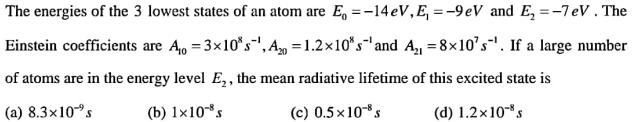
Transcribed Image Text:
The energies of the 3 lowest states of an atom are E = -14 eV, E, = -9 eV and E = -7 eV. The Einstein coefficients are A10 =3x10^8 , A20=1.2x10^8 s' and A21= 8x10^7 s. If a large number of atoms are in the energy level E2, the mean radiative lifetime of this excited state is = The energies of the 3 lowest states of an atom are E = -14 eV, E, = -9 eV and E = -7 eV. The Einstein coefficients are A10 =3x10^8 , A20=1.2x10^8 s' and A21= 8x10^7 s. If a large number of atoms are in the energy level E2, the mean radiative lifetime of this excited state is =
Expert Answer:
Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these physics questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
1 Boltzmann's Epitaph, an idea to die for!?! 2 At the beginning of the 20th century, Ludwig Boltzmann was one of the young physicists championing the particle view of matter. 3 Strange as it may seem...
-
In this problem, we consider the analysis of the combined information from both raters on the shoulder flexion (SF) scores in the posture measurement study. Thus, the questions below concern the data...
-
In which section do you create VLAN on Cisco WLC ? ? Layer 3 3 Section Layer 2 2 Section Security Section RF Section
-
Use of technology, employee retention, and HR development have been at the core of HR becoming more strategic at Xerox. Why have those areas been so key?
-
A Nielsen study indicates that 18- to 34-year olds spend a mean of 93 minutes watching video on their smartphones per week. Source: Data extracted from bit.ly/2rj8GHm. Assume that the amount of time...
-
Find the fundamental natural frequency of a fixed-fixed beam using the static deflection curve \[W(x)=\frac{c_{0} x^{2}}{24 E I}(l-x)^{2}\] where \(c_{0}\) is a constant.
-
Prepare journal entries for the following transactions. Aug. 4 Sold merchandise on account to F. Graves for $340 plus sales tax of 4%, with 2/10, n/30 cash discount terms. 6 Sold merchandise on...
-
Problem #5 (20 points): In an electrostatics experiment two concentric conducting, originally neutral in charge spherical shells are ensembled in a concentric way as described in the figure to the...
-
Three spheres, each of mass m, can slide freely on a frictionless, horizontal surface. Spheres A and B are attached to an inextensible, inelastic cord of length l and are at rest in the position...
-
The CFO of Peaceful Book Binding Company is trying to determine the company's WACC. He has determined that the company's before-tax cost of debt is 8.70%. The company currently has $100,000 of debt,...
-
On December 31, Kohl Company correctly made an adjusting entry to recognize \($2,000\) of accrued salaries payable. On January 8 of the next year, total salaries of \($3,800\) were paid. Assuming the...
-
Even a spring without a block hanging on the end of it has an oscillation period. Why isn't the period zero?
-
Calculate the Lorentz factor for an object moving relative to Earth at (a) \(60 \mathrm{mi} / \mathrm{h}\), (b) \(0.34 \mathrm{~km} / \mathrm{s}\) (speed of sound in air), (c) \(28 \times 10^{3}...
-
A table outfitted with springs on its feet bounces vertically in simple harmonic motion. A cup of coffee is sitting on the table. Discuss whether any of the following changes to this system could...
-
A \(5.0-\mathrm{kg}\) object is suspended from the ceiling by a strong spring, which stretches \(0.10 \mathrm{~m}\) when the object is attached. The object is lifted \(0.050 \mathrm{~m}\) from this...
-
Given the following items from the balance sheet of the Pipper Company: Assets Cash Accounts Receivable Inventory Land Equipment Total Assets Working Capital is: $10,000 15,000 30,000 20,000 25,000...
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
In 2012, Lou has a salary of $54,000 from her job. She also has interest income of $1,700. Lou is single and has no dependents. During the year, Lou sold silver coins held as an investment for a...
-
Van makes an investment in a partnership in 2012. Van's capital contributions to the partnership consisted of $30,000 cash and a building with an adjusted basis of$70,000, subject to a nonrecourse...
-
On February 2, 2012, Alexandra purchases a personal computer for her home. The computer cost $3,000. Alexandra uses the computer 80 percent of the time in her accounting business, 10 percent of the...
-
As the marketing manager for Independent Packaging Pty Ltd, you have asked the accountant what it costs to make the Container2000 model because you want to set a price for the container. A similar...
-
Incomplete information concerning the financial performance of two manufacturing companies is presented below. Required Determine the answers to (a) to(f) for the two companies. Work in process,...
-
Smart Manufacturing Systems Pty Ltd uses a periodic inventory system and closes its accounts on 30 June each year. The companys closing entries made on 30 June 2025 were as shown below. Required (a)...

Study smarter with the SolutionInn App



