The equilibrium constant, K, for the following reaction is 1.2010-2 at 500 K. PCI5(g) =PCl;(g)+ Cl2(g)...
Fantastic news! We've Found the answer you've been seeking!
Question:
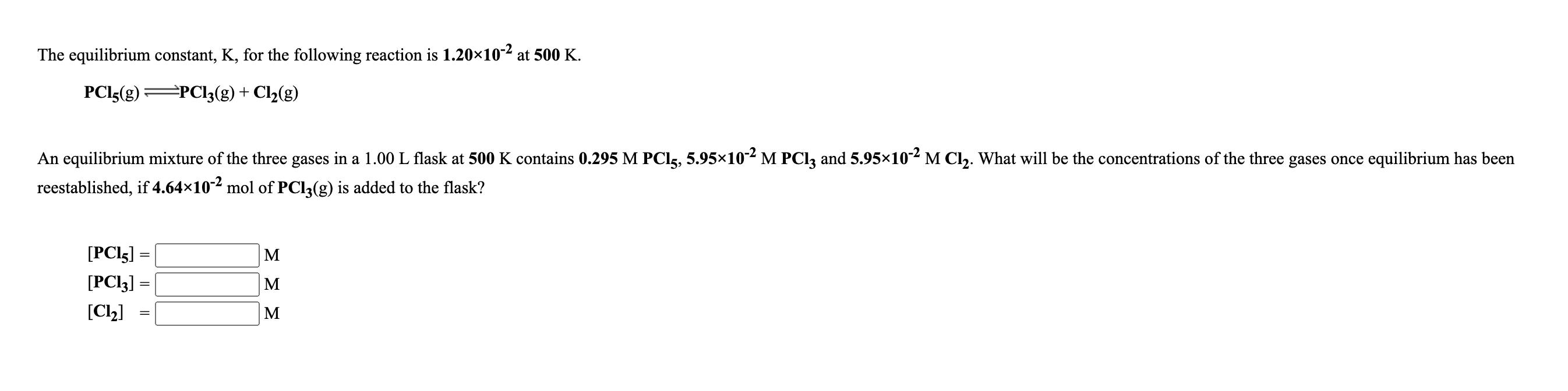
Transcribed Image Text:
The equilibrium constant, K, for the following reaction is 1.20×10-2 at 500 K. PCI5(g) =PCl;(g)+ Cl2(g) An equilibrium mixture of the three gases in a 1.00 L flask at 500 K contains 0.295 M PCI5, 5.95×10- M PCI3 and 5.95×10 M Cl. What will be the concentrations of the three gases once equilibrium has been reestablished, if 4.64x10-2 mol of PCI3(g) is added to the flask? [PCI5] M [PCI3] = M [C2] M The equilibrium constant, K, for the following reaction is 1.20×10-2 at 500 K. PCI5(g) =PCl;(g)+ Cl2(g) An equilibrium mixture of the three gases in a 1.00 L flask at 500 K contains 0.295 M PCI5, 5.95×10- M PCI3 and 5.95×10 M Cl. What will be the concentrations of the three gases once equilibrium has been reestablished, if 4.64x10-2 mol of PCI3(g) is added to the flask? [PCI5] M [PCI3] = M [C2] M
Expert Answer:
Answer rating: 100% (QA)
Given The equilibrium constant K for the following reaction i... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The equilibrium constant K for the reaction 2Cl(g) Cl2(g) was measured as a function of temperature (in kelvins). A graph of ln(K) versus 1/ T for this reaction gives a straight line with a slope of...
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Fe3 + (aq) + Cu(s) Fe2+(aq) + Cu2+(aq) The equation is not balanced.
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Sn4+(aq) + 2Hg(l Sn2+(aq) + Hg2 2+(aq) The equation is not balanced.
-
In Problems 2734, the given pattern continues. Write down the nth term of a sequence {a n } suggested by the pattern. 2 1 1 ,3,-, 5, 7 6 ....: 8
-
Hogan's Consulting showed the following trial balances for its first year just ended December 31, 2014: Required Use the information provided to complete an income statement, statement of changes in...
-
A 15-m-long garden hose has an inner diameter of \(2.5 \mathrm{~cm}\). One end is connected to a spigot; \(20^{\circ} \mathrm{C}\) water flows from the other end at a rate of \(1.2 \mathrm{~L} /...
-
The Wide World of Fluids article titled "Motorized Surfboard,". The thrust to propel the powered surfboard shown in Fig. P5.76 is a result of water pumped through the board that exits as a high-speed...
-
1. Is the pressure for Richard Branson to change Virgins business strategy coming from the internal or external environment? 2. In order to better compete with its rivals, should Virgin focus on a...
-
Which sequences of paths are explored by BFS and DFS in this problem? Exercise 2 Given the graph represented in t Figure 1, where n, is the initial state, n, is the final state, each edge nn' is...
-
s.pdf w19as3sol.doc.pdf 49f233c38bee8583... Create account Login Balance Sheet for a Simplified Bank Assets Cash Gov. Securities $ 100,000.00 190,476.19 Fixed Rate Mortgage Loans 600,000.00 Fixed...
-
How many times does the following code iterate through the while loop? int total = 20; int index = 1; while (index < total) { } if ((total > 9) || (index / total == 5)) { total -= index; } total -=...
-
Write a Java program to determining a leap year: Determining Leap Year? This program first prompts the user to enter a year as an int value and checks if it is a leap year. A year is a leap year if...
-
Thomas Iverson was a founder of CH2O, Inc., and served as the company's president and chairman of the board. CH2O ships blended chemicals to its customers in drums. CH2O asked its customers to return...
-
The following information pertains to the first year of operation for Crystal Cold Coolers Inc.: Number of units produced Number of units sold Unit sales price Direct materials per unit Direct labor...
-
The community nursing service team provides nursing care to patients in the community, Discuss?
-
Solve sin(x) = 0.1 on 0 < x < 2 There are two solutions, A and B, with A
-
Bonds issued by Sizzle Cleaners were priced at $1,096.00 six months ago. The bonds pay semi-annual coupons, have a coupon rate of 7.37%, just made a coupon payment, and have a face value of...
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
Give an example of hydrogen as (a) An oxidizing agent and (b) A reducing agent.
-
Barium metal crystallizes in a body-centered cubic lattice (the Ba atoms are at the lattice points only). The unit cell edge length is 502 pm, and the density of the metal is 3.50 g/cm3. Using this...
-
Predict whether the following solutions are acidic, basic, or nearly neutral: (a) NaBr, (b) K2SO3, (c) NH4NO2, (d) Cr(NO3)3.
-
If a product is produced in high volume by a few large organizations, what phase of the product life cycle is it most likely in?
-
Is a highly standardized product usually produced through a make-to-order or a make-to-stock processing strategy?
-
Does providing customized products generally involve less capital investment or less skilled labor, when compared to more standardized products?

Study smarter with the SolutionInn App


