The formation of molecular hydrogen at 3000 K proceeds through the following elementary reaction: 2H (g)...
Fantastic news! We've Found the answer you've been seeking!
Question:

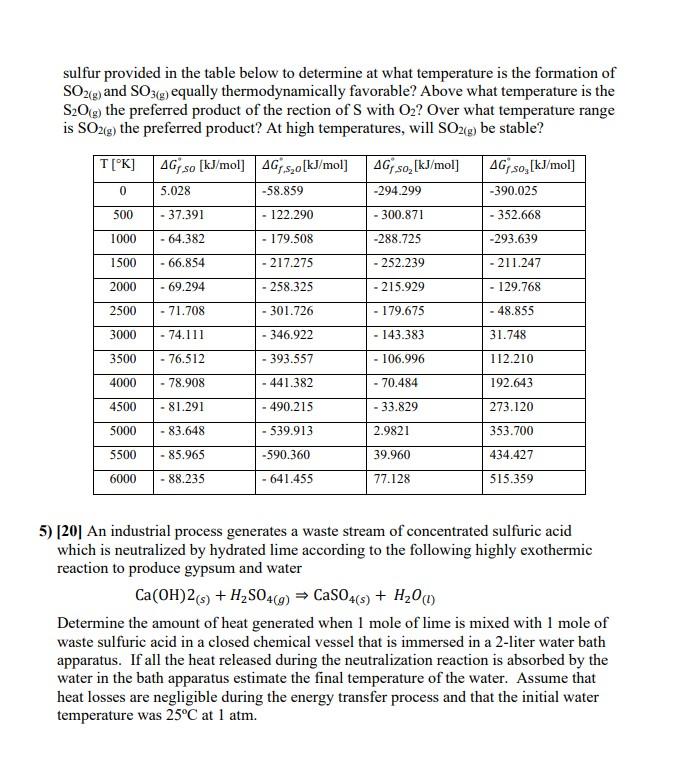
Transcribed Image Text:
The formation of molecular hydrogen at 3000 °K proceeds through the following elementary reaction: 2H (g) + Ar (9) H2(g) + Ar (9) k = 2.2 × 108 mol-2.12.s-1 k, 2.2 x 10 mol-1.L.s-1 Write the rate law for this process with respect to H2. (a) Calculate the rate of the reverse reaction (r=-d[H2(g)]/dt) given [H2(g)] = 4.1×103 mol·L¹ and [Ar(g)] = 4.1×10+ mol·L¹; (b) What concentration of Ar is needed to double the reverse reaction rate computed in (a); (c) Calculate the concentration of [H(g)] needed for the forward reaction rate (r=d[H2(g)]/dt) to be equal to the reverse reaction rate computed under the process conditions given in (a). 2) [20] The decomposition of dinitrogen pentoxide at low temperatures results in the formation of a brown-colored gas with the chemical formula N2O4, known as dinitrogen tetroxide, a harmful pollutant found in the air together with nitrogen dioxide. On the basis of the chemical equation 2N205(g) → 2N204(g) + O2(g) a student assumed that the reaction would be second order. Using the following data, determine whether or not the student's assumption is correct. t/s 0 1200 2400 3600 4800 6000 7200 8400 9600 10800 12000 13000 P(N:Os)/(torr) 268.7 247.2 236.2 227.1 217.8 209.5 201.8 193.2 185.8 178.1 164.9 152.4 3) [10] A piston-cylinder system is used to store 1000 J of work by keeping an ideal gas compressed at a pressure of 4 atm. The stored energy is to be released to the surroundings by allowing the piston to expand isothermally at 25°C to a lower pressure of 1.5 atm. Calculate the amount of gas necessary to carry out the energy transfer if the external pressure is 1 atm. 4) [20] Sulfur oxides are pollutants that contribute to the formation of acid rain, as well as particulate pollution. Use the data on the free energy of formation of the oxides of sulfur provided in the table below to determine at what temperature is the formation of SO2(g) and SO3(g) equally thermodynamically favorable? Above what temperature is the S2O(g) the preferred product of the rection of S with O2? Over what temperature range is SO2(g) the preferred product? At high temperatures, will SO2(g) be stable? AG so [kJ/mol] AGs₂o [kJ/mol] T[°K] 0 5.028 -58.859 500 -37.391 - 122.290 AG so₂ [kJ/mol] -294.299 AG so,[kJ/mol] -390.025 -300.871 - 352.668 1000 - 64.382 - 179.508 1500 - 66.854 -217.275 -288.725 -252.239 2000 - 69.294 - 258.325 - 215.929 2500 - 71.708 -301.726 - 179.675 -293.639 -211.247 - 129.768 - 48.855 3000 - 74.111 -346.922 -143.383 31.748 3500 - 76.512 -393.557 - 106.996 112.210 4000 - 78.908 -441.382 -70.484 192.643 4500 -81.291 -490.215 -33.829 273.120 5000 - 83.648 -539.913 2.9821 353.700 5500 - 85.965 -590.360 39.960 434.427 6000 - 88.235 -641.455 77.128 515.359 5) [20] An industrial process generates a waste stream of concentrated sulfuric acid which is neutralized by hydrated lime according to the following highly exothermic reaction to produce gypsum and water Ca(OH) 2 (s) + H2SO4(9) CaSO4(s) + H2O(1) Determine the amount of heat generated when I mole of lime is mixed with 1 mole of waste sulfuric acid in a closed chemical vessel that is immersed in a 2-liter water bath apparatus. If all the heat released during the neutralization reaction is absorbed by the water in the bath apparatus estimate the final temperature of the water. Assume that heat losses are negligible during the energy transfer process and that the initial water temperature was 25°C at 1 atm. The formation of molecular hydrogen at 3000 °K proceeds through the following elementary reaction: 2H (g) + Ar (9) H2(g) + Ar (9) k = 2.2 × 108 mol-2.12.s-1 k, 2.2 x 10 mol-1.L.s-1 Write the rate law for this process with respect to H2. (a) Calculate the rate of the reverse reaction (r=-d[H2(g)]/dt) given [H2(g)] = 4.1×103 mol·L¹ and [Ar(g)] = 4.1×10+ mol·L¹; (b) What concentration of Ar is needed to double the reverse reaction rate computed in (a); (c) Calculate the concentration of [H(g)] needed for the forward reaction rate (r=d[H2(g)]/dt) to be equal to the reverse reaction rate computed under the process conditions given in (a). 2) [20] The decomposition of dinitrogen pentoxide at low temperatures results in the formation of a brown-colored gas with the chemical formula N2O4, known as dinitrogen tetroxide, a harmful pollutant found in the air together with nitrogen dioxide. On the basis of the chemical equation 2N205(g) → 2N204(g) + O2(g) a student assumed that the reaction would be second order. Using the following data, determine whether or not the student's assumption is correct. t/s 0 1200 2400 3600 4800 6000 7200 8400 9600 10800 12000 13000 P(N:Os)/(torr) 268.7 247.2 236.2 227.1 217.8 209.5 201.8 193.2 185.8 178.1 164.9 152.4 3) [10] A piston-cylinder system is used to store 1000 J of work by keeping an ideal gas compressed at a pressure of 4 atm. The stored energy is to be released to the surroundings by allowing the piston to expand isothermally at 25°C to a lower pressure of 1.5 atm. Calculate the amount of gas necessary to carry out the energy transfer if the external pressure is 1 atm. 4) [20] Sulfur oxides are pollutants that contribute to the formation of acid rain, as well as particulate pollution. Use the data on the free energy of formation of the oxides of sulfur provided in the table below to determine at what temperature is the formation of SO2(g) and SO3(g) equally thermodynamically favorable? Above what temperature is the S2O(g) the preferred product of the rection of S with O2? Over what temperature range is SO2(g) the preferred product? At high temperatures, will SO2(g) be stable? AG so [kJ/mol] AGs₂o [kJ/mol] T[°K] 0 5.028 -58.859 500 -37.391 - 122.290 AG so₂ [kJ/mol] -294.299 AG so,[kJ/mol] -390.025 -300.871 - 352.668 1000 - 64.382 - 179.508 1500 - 66.854 -217.275 -288.725 -252.239 2000 - 69.294 - 258.325 - 215.929 2500 - 71.708 -301.726 - 179.675 -293.639 -211.247 - 129.768 - 48.855 3000 - 74.111 -346.922 -143.383 31.748 3500 - 76.512 -393.557 - 106.996 112.210 4000 - 78.908 -441.382 -70.484 192.643 4500 -81.291 -490.215 -33.829 273.120 5000 - 83.648 -539.913 2.9821 353.700 5500 - 85.965 -590.360 39.960 434.427 6000 - 88.235 -641.455 77.128 515.359 5) [20] An industrial process generates a waste stream of concentrated sulfuric acid which is neutralized by hydrated lime according to the following highly exothermic reaction to produce gypsum and water Ca(OH) 2 (s) + H2SO4(9) CaSO4(s) + H2O(1) Determine the amount of heat generated when I mole of lime is mixed with 1 mole of waste sulfuric acid in a closed chemical vessel that is immersed in a 2-liter water bath apparatus. If all the heat released during the neutralization reaction is absorbed by the water in the bath apparatus estimate the final temperature of the water. Assume that heat losses are negligible during the energy transfer process and that the initial water temperature was 25°C at 1 atm.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these finance questions
-
The expected rate of return of an equity index fund is 12% and the standard deviation of return of this fund is 25%. The rate of a Treasury bill is 4% in the market. You have $2,000,000 investment...
-
Density is not routinely measured in the atmosphere. Why is this not a problem for atmospheric scientists?
-
Assume that in problem 77, Stan sells the machinery in 2022 for $28,500. Determine the effect of the sale on Stan's regular taxable income and his alternative minimum taxable income in 2022. In...
-
Evelyn is the president and sole shareholder of Ephron Corporation. Ephron is an accrual basis taxpayer and uses a calendar year. Evelyn is a cash basis, calendar year taxpayer. During the second...
-
The codon change (Gly-12 to Val-12) in human H-ras that converts it to oncogenic H-ras has been associated with many types of cancers. For this reason, researchers would like to develop drugs to...
-
Maquoketa Services was formed on May 1, 2017. The following transactions took place during the first month. Transactions on May 1: 1. Jay Bradford invested $40,000 cash in the company, as its sole...
-
Prove that if M is a dense linear subspace of a separable Hilbert space H, then H has an orthonormal basis consisting of elements in M. Does the same result hold for arbitrary dense subsets of H?
-
P6. (6 pts) Consider a generator of a CRC scheme 1010011 (i.e., x6+x++x+1). The message is 00110011 10001010. Answer the following questions. a. Compute the CRC bits for this packet (ignoring all...
-
1. List the steps of blood flow through the lungs (pulmonary circulation) starting from the right ventricle to left atrium(5steps) 2. List the steps of blood flow through the heart starting from the...
-
Suppose a major storm takes the pipeline for gasoline "offline" for a short period of time. During that time, the price of gasoline sold increased 30%. Some will label this as "price gouging". Using...
-
Summarize the case study given below Winning the Sydney to Hobart- A Case Study in Project Management Lynn Crawford, University of Technology, Sydney PMI Proceedings, 1993, pp. 53-59 INTRODUCTION...
-
1. Who is required to prepare consolidation accounts? 2. When is the preparation of consolidated accounts not required? 3. When would goodwill on consolidation be considered impaired?
-
"How has business communication has changed post pandemic? Are we changing back or changing to new ways?" 1 source with in text citation and please add the source used
-
Task 2 Prepare complex broking options You are required to prepare a full report for Bill and John by outlining the application process and the risks (potential and real) of which they should be...
-
QUESTION 12 How many moles of sodium bromide can be produced from the reaction of 1.03 moles of sodium with 0.650 moles of bromine gas? How many moles of sodium bromide can be produced from the...
-
The following information is available for Partin Company: Sales $598,000 Sales Returns and Allowances 20,000 Cost of Goods Sold 398,000 Selling Expense 69,000 Administrative Expense 25,000 Interest...
-
Can a firm sustain its operations by maximizing stockholders wealth at the expense of other stakeholders ?
-
In an effort to better understand how her investments are affected by market factors, Michelle Delatorre, the professional tennis player introduced in the Integrative Problem in Chapter, has posed...
-
What type of information can investors get from stock and bond quotation?
-
True or False: Annual worth analysis is the most popular DCF measure of economic worth.
-
Consider a palletizer at a bottling plant that has a first cost of \($150,000,\) operating and maintenance costs of \($17,500\) per year, and an estimated net salvage value of \($25,000\) at the end...
-
True or False: Unless non-monetary considerations dictate otherwise, choose the mutually exclusive investment alternative that has the greatest annual worth over the planning horizon.

Study smarter with the SolutionInn App


