The figure above shows a molecular energy-level diagram. 4 part question (A,B,C,D) Please answers in nm and
Question:
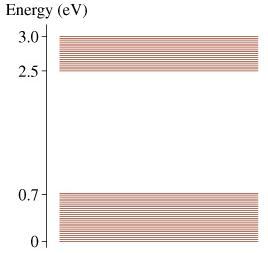
The figure above shows a molecular energy-level diagram. 4 part question (A,B,C,D) Please answers in nm and explain formula thanks!Part A What is the longest wavelength in the molecule?s absorption spectrum?= ? nmPart BWhat is the shortest wavelength in the molecule?s absorption spectrum?= ? nmPart C ( I tried 2486 nm and 1776 nm but was incorrect)What is the longest wavelength in the molecule?s fluorescence spectrum?= ? nmPart D ( I tried 413 nm, but it was incorrect 12400/3.0= 4133.33)What is the shortest wavelength in the molecule?s fluorescence spectrum?= ? nmKeyH= planks constant 6.63 * 10^-34C= speed of light 3*10^8I tried two formulas1. H multiply C divided by differences in Ev2. H multiply C divided by difference in Ev multiply by 1. x 10^-19

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward




