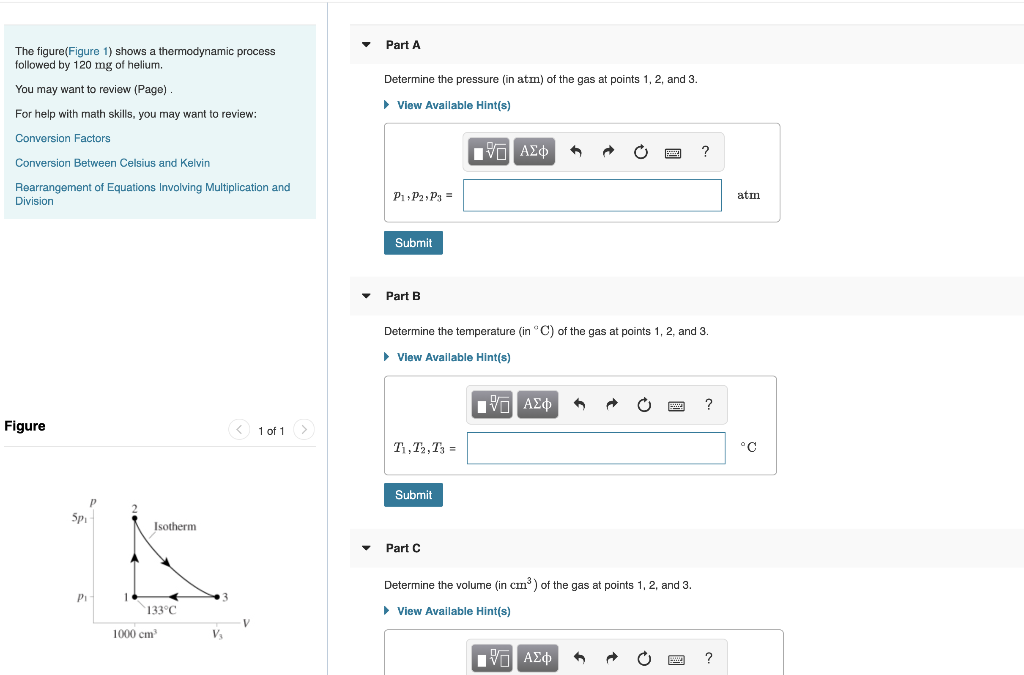
The figure(Figure 1) shows a thermodynamic process followed by 120 mg of helium. You may want...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
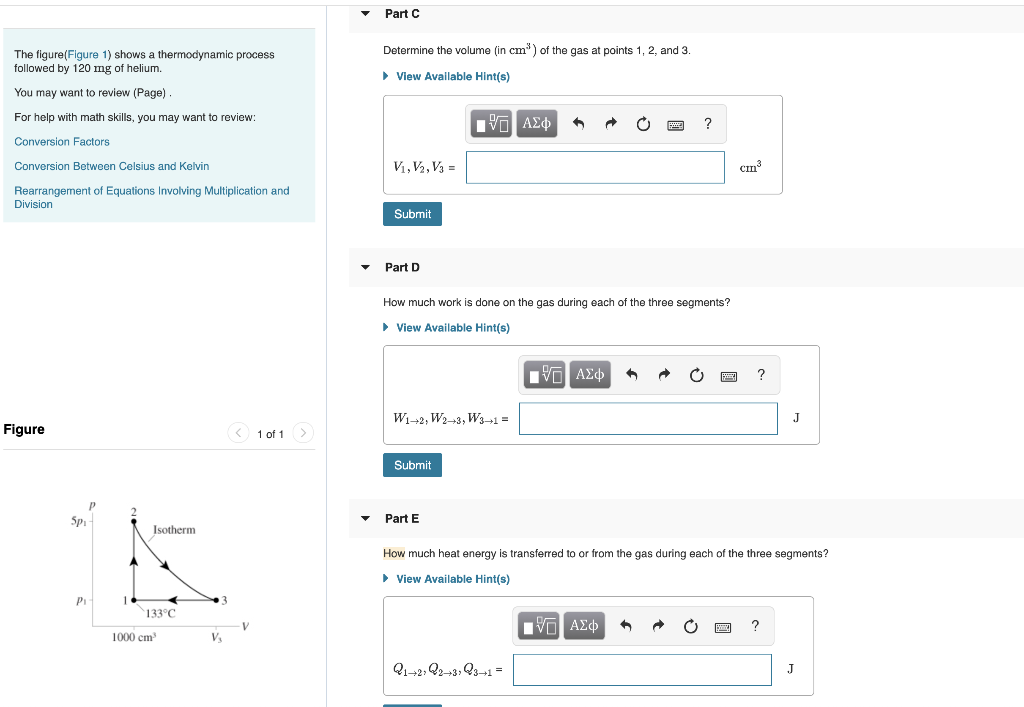
The figure(Figure 1) shows a thermodynamic process followed by 120 mg of helium. You may want to review (Page). For help with math skills, you may want to review: Conversion Factors Conversion Between Celsius and Kelvin Rearrangement of Equations Involving Multiplication and Division Figure Spi P₁ P 1 Isotherm 133°C 1000 cm³ V₂ V 1 of 1 Part A Determine the pressure (in atm) of the gas at points 1, 2, and 3. ► View Available Hint(s) IVE ΑΣΦ P₁ P2, P3= Submit Part B T1, T2, T3 = Determine the temperature (in °C) of the gas at points 1, 2, and 3. ► View Available Hint(s) Submit Part C [IV] ΑΣΦ 6 Determine the volume (in cm³) of the gas at points 1, 2, and 3. ► View Available Hint(s) LG ΑΣΦ ? ? wwwww. ? atm °C The figure(Figure 1) shows a thermodynamic process followed by 120 mg of helium. You may want to review (Page). For help with math skills, you may want to review: Conversion Factors. Conversion Between Celsius and Kelvin Rearrangement of Equations Involving Multiplication and Division Figure Sp P₁- Isotherm 133°C 1000 cm³ V₂ V 1 of 1 Part C Determine the volume (in cm³) of the gas at points 1, 2, and 3. ► View Available Hint(s) V₁, V2, V3 = Submit Part D IVE ΑΣΦ W1+2, W2+3, W3+1 = Submit How much work is done on the gas during each of the three segments? ► View Available Hint(s) Part E ^ 21+22+31 3+1 = IVE ΑΣΦ A PREBE! ? IVE ΑΣΦ 1 cm³ ? How much heat energy is transferred to or from the gas during each of the three segments? ▸ View Available Hint(s) J ? J The figure(Figure 1) shows a thermodynamic process followed by 120 mg of helium. You may want to review (Page). For help with math skills, you may want to review: Conversion Factors Conversion Between Celsius and Kelvin Rearrangement of Equations Involving Multiplication and Division Figure Spi P₁ P 1 Isotherm 133°C 1000 cm³ V₂ V 1 of 1 Part A Determine the pressure (in atm) of the gas at points 1, 2, and 3. ► View Available Hint(s) IVE ΑΣΦ P₁ P2, P3= Submit Part B T1, T2, T3 = Determine the temperature (in °C) of the gas at points 1, 2, and 3. ► View Available Hint(s) Submit Part C [IV] ΑΣΦ 6 Determine the volume (in cm³) of the gas at points 1, 2, and 3. ► View Available Hint(s) LG ΑΣΦ ? ? wwwww. ? atm °C The figure(Figure 1) shows a thermodynamic process followed by 120 mg of helium. You may want to review (Page). For help with math skills, you may want to review: Conversion Factors. Conversion Between Celsius and Kelvin Rearrangement of Equations Involving Multiplication and Division Figure Sp P₁- Isotherm 133°C 1000 cm³ V₂ V 1 of 1 Part C Determine the volume (in cm³) of the gas at points 1, 2, and 3. ► View Available Hint(s) V₁, V2, V3 = Submit Part D IVE ΑΣΦ W1+2, W2+3, W3+1 = Submit How much work is done on the gas during each of the three segments? ► View Available Hint(s) Part E ^ 21+22+31 3+1 = IVE ΑΣΦ A PREBE! ? IVE ΑΣΦ 1 cm³ ? How much heat energy is transferred to or from the gas during each of the three segments? ▸ View Available Hint(s) J ? J
Expert Answer:
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Posted Date:
Students also viewed these physics questions
-
How much work is done on the steam when 1.00 mol of water at 100C boils and becomes 1.00 mol of steam at 100C at 1.00 atm pressure? Assuming the steam to behave as an ideal gas, determine the change...
-
How much work is done on the bowstring of Example 6.9 to draw it back by 20.0 cm? [Rather than recalculate from scratch, use proportional reasoning.]
-
How much work is done by an applied force that moves two charges of 6.5C that are initially very far apart to a distance of 4.5 cm apart?
-
Prepare journal entries for each of the following transactions: 1. Purchase equipment in exchange for cash of $22,400. 2. Provide services to customers and receive cash of $5,100. 3. Pay the current...
-
Smith Law Firm specializes in the preparation of wills for estate planning. On October 1, 2015, the company begins operations by issuing stock for $11,000 and obtaining a loan from a local bank for...
-
The condensed financial statements of Murawski Company for the years 2014 and 2015 are presented shown below. (Amounts in thousands.) Compute the following ratios for 2015 and 2014. (a) Current...
-
In 2014, Air Asia Flight No. 8501 crashed in the Java Sea while flying from Indonesia to Singapore. The crash resulted in the deaths of all passengers and crew on board. The plane involved in the...
-
Stahl Inc. produces three separate products from a common process costing $100,000. Each of the products can be sold at the split-off point or can be processed further and then sold for a higher...
-
3. If the policy proposed in question 2 is adopted, analyze the potential effects of this policy on the demand for workers under the age of 65. That is, if employers who want their employees to be...
-
Trace or copy the graph of the given function f. (Assume that the axes have equal scales.) Then use the method of Example 1 to sketch the graph of f' below it. yA
-
Problem 5 What is the charge (magnitude and sign) of a 2 gram particle if it is stationary in a downward electric field of magnitude 650 [N/C]? E = (650 [N/C])(-) FE F = (mg) (-y) 8
-
elix is considering the purchase of a Porsche Boxster and has negotiated a final price of $48,100. He's trying to decide whether to lease or purchase the vehicle. If he leases, he'll have to pay a...
-
Considering a limit order to buy at the price of $30 with discretionary amount of $0.04, when does the order first execute (if at all)? Time Bid Offer 11:08 30.00 30.05 11:09 30.00 30.04 11:10 30.04...
-
Suppose the risk-free rate is 3.78% and an analyst assumes a market risk premium of 7.37%. Firm A just paid a dividend of $1.47 per share. The analyst estimates the of Firm A to be 1.38 and...
-
Considering a mid-pegged order to buy submitted at 11:00 and the market condition as summarized in below table, when is the order repriced? Time Bid Offer 11:00 52.00 52.02 11:01 52.00 52.02 11:02...
-
A firm earned $484 million in 2021 on a beginning book value of common equity of $2,600 million. The firm paid no dividends and had no stock transactions during that year. It has a required return...
-
Knowing that the lengths X, Y and Z in the figure below are 2 m, 4 m and 2 m, respectively. The length of cable AB in m is: X FAB= 200 N B Y FAC=240 N C Xx Z
-
Figure displays a 12.0 V battery 3 four uncharged capacitors of capacitances C1 = 4.00F, C2 = 6.00F, and C3 = 3.00F. The switch is thrown to the left side until capacitor 1 is fully charged. Then the...
-
What are (a) W out and Q H (b) The thermal efficiency for the heat engine shown in FIGURE EX21.13? p (kPa) 400 Q = -90 J 200 Q =-25 J V (cm) 0- 100 200 FIGURE EX21.13
-
Astronauts on the first trip to Mars take along a pendulum that has a period on earth of 1.50 s. The period on Mars turns out to be 2.45 s. What is the free-fall acceleration on Mars?
-
What is the speed of a proton that has been accelerated from rest through a potential difference of -1000 V?
-
Determine whether each of the following items is an: (a) Cash (b) Dividend to shareholders (c) Land (d) Accounts payable (e) Capital stock (f) Notes payable (g) Accounts receivable (h) Salaries (i)...
-
Assuming the expectations theory is the correct theory of the term structure, calculate the interest rates in the term structure for maturities of one to four years, and plot the resulting yield...
-
Assuming the expectations theory is the correct theory of the term structure, calculate the interest rates in the term structure for maturities of one to four years, and plot the resulting yield...

Study smarter with the SolutionInn App



