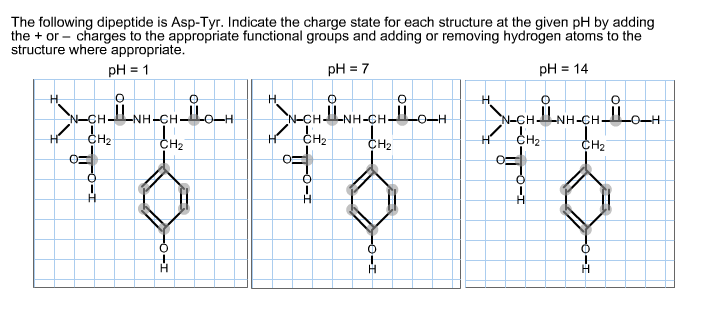
The following dipeptide is Asp-Tyr. Indicate the charge state for each structure at the given pH...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The following dipeptide is Asp-Tyr. Indicate the charge state for each structure at the given pH by adding the + or charges to the appropriate functional groups and adding or removing hydrogen atoms to the structure where appropriate. pH = 1 pH = 7 pH = 14 H 0 NCHINH CHILon H CH₂ A H 0 N-CHINH-CHIL H1 CH₂ 0 0 A -O-H N_CHILNH-CH_II на Енг CH₂ 0 ملی The following dipeptide is Asp-Tyr. Indicate the charge state for each structure at the given pH by adding the + or charges to the appropriate functional groups and adding or removing hydrogen atoms to the structure where appropriate. pH = 1 pH = 7 pH = 14 H 0 NCHINH CHILon H CH₂ A H 0 N-CHINH-CHIL H1 CH₂ 0 0 A -O-H N_CHILNH-CH_II на Енг CH₂ 0 ملی
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Given the following peptide SEPIMAPVEYPK a. Estimate the net charge at pH 7 and at pH 12. Assume the pKa values given in Table 5.1. In Table 5.1 b. How many peptides would result if this peptide were...
-
The condensation of two functional groups can result in the formation of another common functional group, which can be referred to as a compound functional group. Examine the functional groups in...
-
The hydrogen atoms of the g carbon of crotonaldehyde are appreciably acidic (pKa 20). (a) Write resonance structures that will explain this fact. (b) Write a mechanism that accounts for the following...
-
Mr. CFE is the sole proprietor of a hardware store called, The CFE Shop. Mr. CFE has decided to incorporate the business but wishes to minimize any income inclusions on the transfer of his business...
-
Customers arrive to check in at the exclusive and expensive Regency Hotels lobby at a rate of 40 per hour (Poisson distributed). The hotel normally has three clerks available at the desk to check in...
-
Define biological productivity. When a scientist says productivity is a rate, what does she or he mean? Discuss several factors that influence productivity in various ocean environments (i.e., the...
-
What stakeholder affected by their behavior was Gilead weighting very lightly when it decided what to tell the FDA about the medicines it wished to have permission to sell?
-
Balance sheet data for the Dover Hot Tub Company on December 31, the end of the fiscal year, are shown at the top of the following page. Prepare a comparative balance sheet for 2008 and 2007, stating...
-
8. Suppose f(x) = x 2. Use the forward difference method with h = 0.1 to approximate f'(1).
-
1. What is the cost of debt for Sunrise Bakery? 2. What is their cost of equity? 3. What is the WACC? 4. Which cost of capital should be used to evaluate the feasibility of the oven purchase? 5....
-
Consider the three main justifications given for affirmative action at Omega. a. Omega's history b. Omega's workforce is 5 percent African American and 15 percent Hispanic, and the population of...
-
A detailed sales analysis can show a sales manager: a. why sales volumes are down in certain areas? b. where the company should open new branch offices next? c. how to divide up territories? d. how...
-
Bernard Auto has shareholders equity of $121,400. The firm owes a total of $132,000 of which 40% is payable within the next year. The firm has net fixed assets of $151,620. what is the amount of the...
-
A nurse manager is preparing for a performance appraisal review for an employee who has been identified as substandard. describe the process of conducting a performance appraisal review and when...
-
Jessica spent $700 on an online training course to learn how to invest in municipal bonds which generate tax-exempt income. She used $50,000 to purchase the bonds and received interest of $4,000 in...
-
What would be the total cost of leasing a vehicle for four years that requires a security deposit of $1,000 (which would be withdrawn from your portfolio, which earns 9% per year), has monthly lease...
-
iDetail ran a booming IBC business during the winter semester. They would detail automobiles while customers waited. With their initial way of cleaning vehicles--sequentially, like an assembly...
-
Write the binomial probability in words. Then, use a continuity correction to convert the binomial probability to a normal distribution probability. P(x 110)
-
Part 1 a. Both NaCl and MgCl 2 are soluble ionic compounds. Write the balanced chemical equations for these two substances dissolving in water. b. Consider the pictures below. These pictures...
-
A sample of potassium aluminum sulfate 12-hydrate, KAl(SO4)2 12H2O, containing 118.6 mg is dissolved in 1.000 L of solution. Calculate the following for the solution: a. The molarity of KAl(SO4)2....
-
Write the following measurements, without scientific notation, using the appropriate SI prefix. a. 4.851 106 g b. 3.16 102 m c. 2.591 109 s d. 8.93 1012 g
-
The liquidity premium hypothesis also holds that issuers of bonds prefer to issue long-term bonds to lock in borrowing costs. How would this preference contribute to a positive liquidity premium?
-
Go to stockcharts.com/freecharts/yieldcurve.php where you will find a dynamic or living yield curve, a moving picture of the yield curve over time. Hit the Animate button to start the demonstration....
-
Suppose that the price of 3-year zero-coupon bonds is $816.30. What is the forward rate for the third year? How would you construct a synthetic 1-year forward loan that commences at t = 2 and matures...

Study smarter with the SolutionInn App


