The following June transactions relate to KitchenApps Inc. June 6: Purchase of inventory with a list...
Fantastic news! We've Found the answer you've been seeking!
Question:
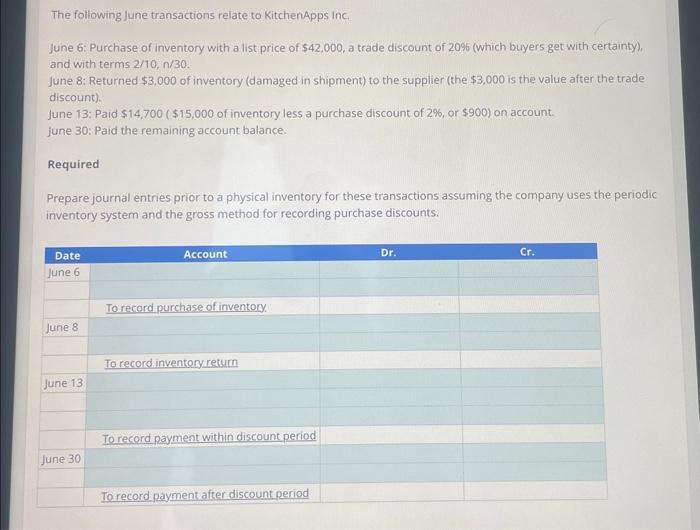
Transcribed Image Text:
The following June transactions relate to KitchenApps Inc. June 6: Purchase of inventory with a list price of $42,000, a trade discount of 20% (which buyers get with certainty), and with terms 2/10, n/30. June 8: Returned $3,000 of inventory (damaged in shipment) to the supplier (the $3,000 is the value after the trade discount). June 13: Paid $14,700 ($15,000 of inventory less a purchase discount of 2%, or $900) on account. June 30: Paid the remaining account balance. Required Prepare journal entries prior to a physical inventory for these transactions assuming the company uses the periodic inventory system and the gross method for recording purchase discounts. Date June 6 June 8 June 13 June 30 Account To record purchase of inventory. To record inventory return To record payment w in discount period To record payment after discount period Dr. Cr. The following June transactions relate to KitchenApps Inc. June 6: Purchase of inventory with a list price of $42,000, a trade discount of 20% (which buyers get with certainty), and with terms 2/10, n/30. June 8: Returned $3,000 of inventory (damaged in shipment) to the supplier (the $3,000 is the value after the trade discount). June 13: Paid $14,700 ($15,000 of inventory less a purchase discount of 2%, or $900) on account. June 30: Paid the remaining account balance. Required Prepare journal entries prior to a physical inventory for these transactions assuming the company uses the periodic inventory system and the gross method for recording purchase discounts. Date June 6 June 8 June 13 June 30 Account To record purchase of inventory. To record inventory return To record payment w in discount period To record payment after discount period Dr. Cr.
Expert Answer:
Answer rating: 100% (QA)
Here are the journal entries for the June transactions of KitchenApps Inc June 6 Inventory ... View the full answer

Related Book For 

Corporate Financial Accounting
ISBN: 978-1133952411
12th edition
Authors: Carl S. Warren, James M. Reeve, Jonathan E. Duchac
Posted Date:
Students also viewed these accounting questions
-
Writing clear, assay succinct procedures (SOPs) can be one important aspect of a Manager's job.Sometimes, technical personal are tasked with procedure writing, but it is the manager's responsibility...
-
The following independent situations relate to inventory accounting: 1. Draper Co. purchased goods with a list price of $175,000 and a trade discount of 20% based on the quantity purchased, with...
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Using examples outline the main advantages and disadvantages of one of the following models of corporate governance: i) The principal/agent model ii) The stakeholder mode
-
A circular coil of wire has a radius of 7.5 cm and has 125 turns of wire that carries 6.6 A of current. What is the magnetic field at the center of the coil?
-
APCO Company manufactures various lines of bicycles. Because of the high volume of each line, the company employs a process cost system using the weighted-average method to determine unit costs....
-
In Exercises 1518, use the given set of points to a. Compute b1. b. Compute the residual standard deviation se. c. Compute the sum of squares for x, (x x)2. d. Compute the standard error of b1, sb....
-
The adjusted trial balance of Sang Company shows the following data pertaining to sales at the end of its fiscal year October 31, 2017: Sales Revenue $820,000, Freight-Out $16,000, Sales Returns and...
-
There are multiple pieces required for this week's assignment, so please be sure to address each one. Discuss all the factors that contributed to the growth of the private security industry,...
-
A new company is planning to make and sell new cheap smart phones in America. They will sell the phone at $99.99 in a market that potentially buys 1 million smart phones every year. They hope to get...
-
Quantity supplied 7,200 Price, $ Quantity demanded 210 800 1.80 1,00 4,800 1.60 2,400 2,400 140 3,200 800 1.20 4.100 200 The table above shows the demand and supply schedule for a football ticket....
-
Acme Company has two divisions, East Division and West Division. Here are data regarding the current year: East Division West Division Sales revenue $560,000 $478,500 Variable costs 230,000 231,000...
-
Nelson Company reported cost of goods sold of $720,000 last year and $750,000 this year. Nelson also reported accounts payable of $260,000 last year and $240,000 this year. Compute this year's...
-
What is the basic elements of a distributive bargaining situation and integrative negotiation? Explore the strategy and tactics of integrative negotiation with the specific example.
-
Imagine you were a high-level manager for Toby Groves at his company prior to the "air loan" scandal. You earnestly want to save the company, save jobs, and help the company get out of this...
-
Determine an equation for the parabola that has a vertex at the point (1,2) and contains the point (3,-4). Then determine the y-coordinate for the point where the parabola intersects the y-axis.
-
2. Show that in Z, the following equality is satisfied 1 + 2 +2 +2 + 2t +... 3 Ijustify your answer in detail)
-
Before the latest financial crisis and recession, when was the largest recession of the past 50 years, and what was the cumulative loss in output over the course of the slowdown?
-
Selected data derived from the income statement and balance sheet of Jones Soda Co. for a recent year are as follows: Income statement data (in thousands): Net earnings (loss) ...............$(6,106)...
-
Describe the nature of the assets that compose the following sections of a balance sheet: (a) Current assets, (b) Property, plant, and equipment.
-
Describe two reports provided by independent auditors in the annual report to shareholders.
-
The heat of vaporization of ether is \(25.98 \mathrm{~kJ} / \mathrm{mol}\) at its boiling point, \(34.5^{\circ} \mathrm{C}\). (a) Calculate the rate of change of vapour pressure with temperature...
-
Show that the internal energy of an ideal gas is a function only of temperature.
-
For a gas obeying the equation of state \(V=B+\frac{R T}{P}\), the Joule-Thomson coefficient is given by \[ \mu_{\mathrm{JT}}=-\frac{1}{C_{P}}\left(T \frac{d B}{d T}-B ight) \]

Study smarter with the SolutionInn App


