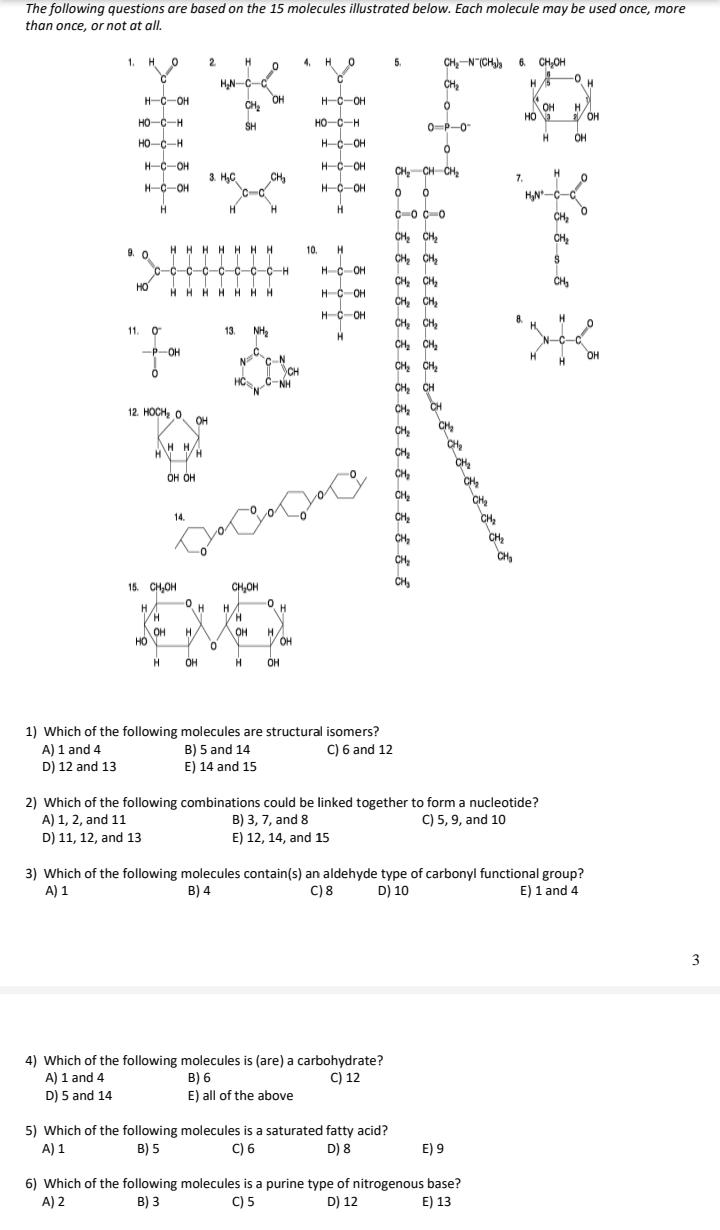
The following questions are based on the 15 molecules illustrated below. Each molecule may be used...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. CH,-N (CH, 6. CH,OH CH 1. H O 2 4. H O 5. 0- HN-C-C OH CH H. H-C-OH H-C-OH OH / OH HO-C-H SH HO-C-H 0-P-0 HO-C-H HC-OH H. OH H-C-OH H-C-OH CH, CH CH, 3. H,C CH 7. H-C-OH HC-OH HN-C H H. H. C-o C-0 CH, CH CH2 CH, HHHHHHH 9. 0 c-c-c-c-c-c-c-c-H 10. CH CH2 HC-OH CH CH, CH, CH, CH, CH, CH, HO CH HHHH HHH HC-OH H-C-OH H. 11. 0 13. CH, CH, CH, CH, CH -P-OH C. N C-N CH C-NH OH 12. HOCH, O CH, CH OH CH, H H/ CH, CH, CH, OH OH CH CH, CH, CH, 14. CH, CH, CH 15. CH,OH CH,OH H. H. H OH HO H. H. V OH OH OH H. OH 1) Which of the following molecules are structural isomers? B) 5 and 14 E) 14 and 15 A) 1 and 4 D) 12 and 13 C) 6 and 12 2) Which of the following combinations could be linked together to form a nucleotide? B) 3, 7, and 8 E) 12, 14, and 15 A) 1, 2, and 11 C) 5, 9, and 10 D) 11, 12, and 13 3) Which of the following molecules contain(s) an aldehyde type of carbonyl functional group? C) 8 A) 1 B) 4 D) 10 E) 1 and 4 4) Which of the following molecules is (are) a carbohydrate? B) 6 E) all of the above C) 12 A) 1 and 4 D) 5 and 14 5) Which of the following molecules is a saturated fatty acid? C) 6 A) 1 B) 5 D) 8 E) 9 6) Which of the following molecules is a purine type of nitrogenous base? C) 5 A) 2 B) 3 D) 12 E) 13 The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. CH,-N (CH, 6. CH,OH CH 1. H O 2 4. H O 5. 0- HN-C-C OH CH H. H-C-OH H-C-OH OH / OH HO-C-H SH HO-C-H 0-P-0 HO-C-H HC-OH H. OH H-C-OH H-C-OH CH, CH CH, 3. H,C CH 7. H-C-OH HC-OH HN-C H H. H. C-o C-0 CH, CH CH2 CH, HHHHHHH 9. 0 c-c-c-c-c-c-c-c-H 10. CH CH2 HC-OH CH CH, CH, CH, CH, CH, CH, HO CH HHHH HHH HC-OH H-C-OH H. 11. 0 13. CH, CH, CH, CH, CH -P-OH C. N C-N CH C-NH OH 12. HOCH, O CH, CH OH CH, H H/ CH, CH, CH, OH OH CH CH, CH, CH, 14. CH, CH, CH 15. CH,OH CH,OH H. H. H OH HO H. H. V OH OH OH H. OH 1) Which of the following molecules are structural isomers? B) 5 and 14 E) 14 and 15 A) 1 and 4 D) 12 and 13 C) 6 and 12 2) Which of the following combinations could be linked together to form a nucleotide? B) 3, 7, and 8 E) 12, 14, and 15 A) 1, 2, and 11 C) 5, 9, and 10 D) 11, 12, and 13 3) Which of the following molecules contain(s) an aldehyde type of carbonyl functional group? C) 8 A) 1 B) 4 D) 10 E) 1 and 4 4) Which of the following molecules is (are) a carbohydrate? B) 6 E) all of the above C) 12 A) 1 and 4 D) 5 and 14 5) Which of the following molecules is a saturated fatty acid? C) 6 A) 1 B) 5 D) 8 E) 9 6) Which of the following molecules is a purine type of nitrogenous base? C) 5 A) 2 B) 3 D) 12 E) 13
Expert Answer:

Related Book For 

Intermediate accounting
ISBN: 978-0077647094
7th edition
Authors: J. David Spiceland, James Sepe, Mark Nelson
Posted Date:
Students also viewed these chemical engineering questions
-
The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. 1. H O 2 4. H O 5. CH,-N (CH, 6 CH,OH CH 0- HN-C-C OH CH H....
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study property, plant,...
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study property, plant,...
-
What is true about business cycles? Multiple select question. They vary in duration and intensity. They follow an identical pattern. They are made up of alternating rises and declines. "Ups" are...
-
A tax-deferred sale of a business by a reorganization of share capital may present more risk to the vendor than a sale of its shares to a corporate purchaser and an election of a transfer price for...
-
Iron Man Products has projected the following sales for the coming year: Sales in the year following this one are projected to be 15 percent greater in each quarter. a. Calculate payments to...
-
Refer to the statements for Google in Appendix A. For the year ended December 31, 2015, what was its debt-to-equity ratio? What does this ratio tell us? Data From Statement Google In Appendix A...
-
Stratford Company distributes a lightweight lawn chair that sells for $15 per unit. Variable costs are $6 per unit, and fixed costs total $180,000 annually. Required: Answer the following independent...
-
Use the attached image and SQL DDL Commands to query the appropriate tables and populate with four rows of data. Please include explanation for script. CUSTOMER PK CustomerID CustomerName Address...
-
A binary mixture of mole fraction zj is flashed (o conditions T and P, Fur one of the following determine: the equilibrium mole fractions x1 and y1 of the liquid and vapor phases formed, the molar...
-
Answer the Following True/False. Taxpayers who sell or exchange their personal residence may exclude up to $250,000 of gain if it was owned and occupied as a principal residence for at least two...
-
How should endowment investments that are permanent in nature be reported on a university Statement of Net Assets? a. As a noncurrent asset b. As a current or noncurrent asset as per the donors...
-
A nongovernment not-for-profit organization would present all of the following categories of cash flows except a. cash flows from noncapital financing activities. b. cash flows from investing...
-
Which of the following statements is not required to be presented for an Investment Trust Fund? a. Statement of net assets. b. Statement of changes in net assets. c. Statement of cash flows. d. All...
-
Restricted net assets for a governmental college or university are commonly subclassified for a. designated net assets. b. reserved net assets. c. nonexpendable net assets. d. expendable net assets....
-
Which of the following must be prepared for Agency Funds as part of a governments Basic Financial Statements? a. Statement of changes in net assets. b. Statement of revenues, expenditures, and...
-
DA SILVA Plcs shares are not traded in any recognized market. Its sole activity is saloon and car hire. It is financed by a combination of 2 million $0.5 ordinary shares and a $1.5 bank loan. Very...
-
Provide a draft/outline of legal research involving an indigenous Canadian woman charged with assault causing bodily harm under (Sec 267b) of the Criminal Code, where the crown wants a 12-month jail...
-
At the beginning of 2013, a company adopts the dollar-value LIFO inventory method for its one inventory pool. The pools value on that date was $1,400,000. The 2013 ending inventory valued at year-end...
-
At December 31, 2013, the financial statements of Hollingsworth Industries included the following: Net income for 2013 ...................... $560 million Bonds payable,10%, convertible into 36...
-
During 2013, WMC Corporation discovered that its ending inventories reported on its financial statements were misstated by the following amounts: 2011 . understated by . $120,000 2012 . overstated by...
-
Over what ranges of taxable income in 2017 will the total income tax liability for two persons with equal incomes who file as single individuals equal their income tax liability if they file jointly...
-
If a taxpayer has $40,000 of employee salary in 2017, how much will be withheld for the Social Security and Medicare taxes?
-
Sally and Jim are married and have taxable income in 2017 of $160,000. If they could file their income tax as single individuals, each of them would have taxable income of $80,000. Do they have a...

Study smarter with the SolutionInn App


